Bromonium ions can be captured by nucleophiles other than water. Predict the products of each of the
Question:
a.
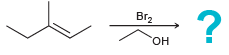
b.
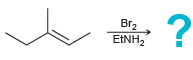
Transcribed Image Text:
Br2 "он Br2 EINH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
a b ...View the full answer

Answered By

Mamba Dedan
I am a computer scientist specializing in database management, OS, networking, and software development. I have a knack for database work, Operating systems, networking, and programming, I can give you the best solution on this without any hesitation. I have a knack in software development with key skills in UML diagrams, storyboarding, code development, software testing and implementation on several platforms.
4.90+
67+ Reviews
156+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the products of each of the following reactions. (a) (b) (c) (d) OH Cl pyridine OH (1) NaH (2) CH2l HBr OH HNOg, H2SO4 H3C
-
Predict the products of each of the following reactions: a. b. c. d. e. f. g. h. i. 1) BH3 THF 2) H2O2, NaOH Pt
-
Under acid-catalyzed conditions, epoxides can be opened by a variety of nucleophiles other than water, such as alcohols. In such a case, the nucleophile will generally attack at the more substituted...
-
8) Implement a class UserNames which requires an ArrayList of names (user names), consider the following static methods methods: initilizeUserName(), deleteUserName(), addUserName() and display...
-
What is lordosis and how does it relate to a lumbar pad?
-
You are asked to predict the next item in the following sequence of integers: 1,2,3. If your prediction is 4 , what assumptions did you make?
-
According to Boyle's law for gases (a) \(V \propto T\) while \(P=\) constant (b) \(V \propto 1 / P\) while \(T=\) constant (c) \(V \propto 1 / T\) while \(P=\) constant (d) \(V \propto P\) while...
-
Cool Logos buys logo- imprinted merchandise and then sells it to university bookstores. Sales are expected to be $ 2,005,000 in September, $ 2,250,000 in October, $ 2,381,000 in November, and $...
-
a) Differentiate (with illustration) the differences between non-blocking and blocking message passing operation. (8 marks) b) Describe THREE (3) advantages of threaded programming as compared to...
-
There are 2 shinobis with chakra levels 5 and 10 respectively and the desired sum of chakra levels is utmost 15 Starting with ke0, suy of chakra levels after attack max(5-0,0) + max(10- 0,0) 5+10 15....
-
Draw the mechanism of the following reaction: ONa + NaBr Br
-
For each of the following objects determine whether or not it possesses a plane of symmetry: a. b. c. d. e. f.
-
For the reaction NH3(g) + H2S(g) NH4HS(s) K = 400. at 35.0oC. If 2.00 moles each of NH3, H2S, and NH4HS are placed in a 5.00- L vessel, what mass of NH4HS will be present at equilibrium? What is the...
-
You want to buy a $32,000 car. The company is offering a 4% interest rate for 36 months (3 years). What will your monthly payments be?
-
Last year, your company sold electronic products to Brazil. You are expecting to receive Real 2,000,000 in 6 months. The following quotations are provided by a currency dealer: In $ Real 0.4200 1 m....
-
Bruin, Incorporated, has identified the following two mutually exclusive projects: Year Cash Flow (A) Cash Flow (B) 0 -$ 51,000 -$ 51,000 1 27,000 2 21,000 3 4 16,500 12,200 14,900 18,900 23,000...
-
What is the future value of $35 per week for 30 years at 9.5 percent interest? Assume the first payment occurs at the end of this week.
-
A cylinder 0.1 m diameter rotates in an annular sleeve 0.102 m internal diameter at 100 r.p.m. The cylinder is 0.2 m long. If the dynamic viscosity of the lubricant between the two cylinders is 1.0...
-
Use the results of Exercises 7475 to describe and graph the following circles. r 2 + 2r(cos - 3 sin ) = 4
-
The manager of a local convenience store is expanding his line of small toy items. To price these new items, the manager is looking at the prices being charged by competing retailers in his area. For...
-
Draw all the isomers for each of these formulas. The total number for each is given in parentheses. (a) C3H8O (3) (b) C4H9Cl (4) (c) C4H8 (5) (d) C7H16 (9)
-
The formula C4H8O has many isomers. (a) Draw three isomers that have a carbon oxygen double bond. What functional group is present in each of them? (b) Draw three alcohols with this formula. (c) Draw...
-
Four of the ten isomers of C5H10, are shown in Figure 2.5. Draw four other isomers with this formula. Line structure Condensed Skeletal Molecular o Kekule structure model structure structure ---...
-
Assume that management wants wage concession from its counterpart labor union. Explain why this often leads to disputes. Suppose management gives up some of its future negotiating power by subjecting...
-
Why have you chosen this nursing Field of Specialization or Major? What influenced you in selecting your intended major of study and what goal(s) have you set for the future?
-
What are logical fallacies and how can they affect our decision-making?

Study smarter with the SolutionInn App


