Carefully read the following curved arrows shown and draw the expected alkene that is produced by this
Question:
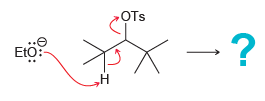
Transcribed Image Text:
OTs -? Eto:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
This me...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using curved arrows, show the principal fragments that would be observed in the mass spectrum of each of the following compounds: a. b. c. d. e. f. CH CH CH CH CH OH CH CCH CH2CH2CH3 CH3CH...
-
Is the current that is produced by a common generator ac or dc?
-
For each of the following cases, read the curved arrows and identify which arrow-pushing pattern is utilized: a. b. c. d. e. f. g. h. i. :Ci: -0-H - 0-H 0:0-I :O:
-
BC Lessee Co. enters a non-cancelable lease agreement with Blackcomb Co. on January 1, 2023, under which Blackcomb Co. leases equipment to BC Lessee Co. Information pertaining to the lease is...
-
During a plastic-limit test, the following data was obtained for one of the samples: Wet weight + container = 23.13 g Dry weight + container = 19.12 g Container weight = 1.50 g What is the PL of the...
-
What does the tax law include in amount realized?
-
Identify the singularity and find the nature of solution near the singularity for van der Pol's equation: \[\ddot{x}-\alpha\left(1-x^{2} ight) \dot{x}+x=0\]
-
Montana Company experienced the following events during 2012. 1. Acquired $30,000 cash from the issue of common stock. 2. Paid $12,000 cash to purchase land. 3. Borrowed $10,000 cash. 4. Provided...
-
Answer the questions in the picture below using all listed information 15. Ugh Kinetic Elastic 200 ZE 2 kg 1 m 2 kg 0.5 A 2 kg mass is pushed 0.5 m into a spring with spring constant 200 N/m on a...
-
State with brief reason whether the following statements are true, false, or uncertain: a. In the presence of heteroscedasticity OLS estimators are biased as well as inefficient. b. If...
-
Describe the general characteristics of each of the following groups of insecticides: chlorinated hydrocarbons, organophosphates, and carbamates.
-
It is more effective to use the sterile male technique when an insect population is small than when it is large. Explain.
-
Determine the reactions for each structure. All dimensions are measured from the centerlines of members. 25 kips 4 8' B 9 kips/ft P = 15 kips C D 8 44 | E
-
Frank's Fluids has a market value of Debt of $5,000,000 and outstanding Equity Valued at $50,000,000. The company has a WACC of 10%. The company's NOPAT is $7,500,000 and its EBITDA is $8,200,000....
-
What are some of the pros and cons of shifting elements of the portfolio, including rebalancing or making investment changes in terms of their tax liability? Showing full detail and specific examples...
-
Given the table below answer the question. 2011 2012 Cash Accounts receivable Inventory 200 100 800 1,400 3,600 3,100 Current assets 4,600 4,600 Gross fixed assets 22,000 31,000 (Accumulated...
-
Jack owns an initial wealth (W0) of $30. Inadvertently, he agreed to enter into the following game : - With 1/4 probability his wealth goes up to $50 - With 1/2 probability his wealth stays the same...
-
Clay Buckley dressed his last game for Duke in Indianapolis in 1991. On a Monday. He placed $45,000 into an account and let it grow at 12% compounded quarterly for 31 years. Approximately how much...
-
Evaluate the expression. C(8, 0)
-
What are the key elements of a system investigation report?
-
Compound A, C7H12, was found to be optically active. On catalytic reduction over a palladium catalyst, 2 equivalents of hydrogen were absorbed, yielding compound B, C7H16. On ozonolysis of A, two...
-
Compound A, C 11 H 16 0, was found to be an optically active alcohol. Despite its apparent unsaturation, no hydrogen was absorbed on catalytic reduction over a palladium catalyst. On treatment of A...
-
One of the steps in fat metabolism is the hydration of crotonate to yield 3-hydroxybutyratc. The reaction occurs by addition of OH to the Si face at C3, followed by Protonation at C2, also from the...
-
You have received an inquiry from a prospective client, Lazy Sofa, Inc., concerning the accounting for investments. Betty Jason, owner of Lazy Sofa, is considering investing some of her companys idle...
-
Firm A invents a new product and obtains a patent for it. It calls the product "Asky". Because of the patent, only Firm A can produce and sell Asky. Firm A plans to sell 1,000 units of Asky at the...
-
You are the Senior Manager of IAuditYou LLP, you were recently assigned to take over a very important client for the company, The engagement partner, Max Roff, has been the audit partner for the past...

Study smarter with the SolutionInn App


