Cinchocaine is a long-acting local anesthetic used in spinal anesthesia. Identify the most basic nitrogen atom in
Question:
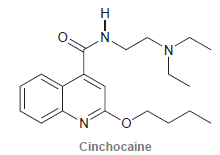
Transcribed Image Text:
Н N. N. Cinchocaine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
I...View the full answer

Answered By

Geoffrey Isaboke
I am an industrious tutor with a 5-yr experience in professional academic writing. I have passion for History and Music and I have good knowledge in Economics
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the structure of lysergic acid diethylamide (LSD), a potent hallucinogen containing three nitrogen atoms. One of these three nitrogen atoms is significantly more basic than the other two....
-
Which is the most basic nitrogen in each compound. Explain your choices. (a) (b) (c) NH2 HN
-
Novocaine, used as a local anesthetic by dentists, is a weak base (Kb = 8.91 10-6). What is the ratio of the concentration of the base to that of its acid in the blood plasma (pH 5 7.40) of a...
-
The annual growth rate in EPS is 3%. The P/E ratio is 12x. EPS last year was $1.00. Calculate What should the stock price be in 4 years?
-
"The information below relates to Milton Company's trading securities in 2012 and 2013. (a) Prepare the journal entries for the following transactions. January 1, 2012 Purchased $300,000 par value of...
-
For a Markov process define its infinitesimal operator: \[T(t) f\left(X_{s} ight)=\int f(y) p\left(t, X_{s}, y ight) d y=E\left[f\left(X_{t+s} ight) \mid X_{s} ight]\] Show that this operator is a...
-
If the majority in a community finds an artistic representation of their religion, race, or culture offensive, should it be censored? The statue of President Theodore Roosevelt flanked by an African...
-
The numbered items that follow are concepts that underlie value-based systems, such as ABM and lean operations. Match each concept to the related lettered element(s) of a lean operating environment....
-
Southwest Milling Company purchased a front-end loader to move stacks of lumber. The loader had a list price of $124,960. The seller agreed to allow a 5.00 percent discount because Southwest Milling...
-
Jennifer Nantale is a new staff auditor on the audit engagement of Greenville Light & Sound, which is a publicly traded company with a calendar year end. The engagement team has completed its review...
-
What is the rule of three? Suppose you worked for a small firm in a large industrysuch as a small manufacturer of furniture. How might you actually use the rule of three to enhance your firms...
-
How does the BCG matrix help marketers decide which products to offer? According to the matrix, which types of products are most desirable, and why?
-
The near point of an eye is 100 cm. A corrective lens is to be used to allow this eye to clearly focus on objects 25.0 in front of it. (a) What should be the focal length of the lens? (b) What is the...
-
Research: By 2000, every state in the United States had adopted whistleblower protection statutes of some type. Locate one such law from Missouri. Under what circumstances are whistleblowers...
-
Part I -Please define and describe the following legal concepts: Reasonable Suspicion Probable Cause Proof Beyond a Reasonable Doubt Part II - Please explain which of these concepts requires the most...
-
A cart with a mass of 67.52 kilograms accelerates from 0.91 to 7.39 meters per second in 3.69 seconds. Find the net force (in newtons) on the cart. Round your answer to two decimal places.
-
(f-g)(x)=x+7-x+6 What is the domain off-g? OA. The domain of f- g is OB. The domain of f -g is O C. The domain of f-g is (Use a comma to separate answers as needed.) (Type your answer in interval...
-
If 27m-n = and 49m2n = 7, the values of m and n are
-
Evaluate the following integrals. et dx e* 2e*
-
One study found that the elderly who do not have children dissave at about the same rate as the elderly who do have children. What might this finding imply about the reason the elderly do not dissave...
-
Show how the product of part (a) can be converted into estrone. Me O HIH HO estrone
-
In 1985, two researchers at the University of California, Riverside, carried out the reaction given in Fig. P27.51. The equilibrium mixture contained compound A (22%), a single stereoisomer of B...
-
(a) What type of pericyclic reaction is required to form benzene from Dewar benzene? (b) Explain why Dewar benzene, although a very un-stable molecule, is not readily transformed to benzene....
-
Company XYZ is considering investing in a new machine that will cost $15,000. The machine will allow the firm to generate sales of $50,000 per year. The machine has a useful life of three years....
-
Dareka's Investments Limited (DIL) is considering two projects in Central Jamaica. The initial capital outlay for each project is US $45,000 and the cost of capital for the DIL is 12%. The cash flow...
-
Calculate the price of a three-month European put option on a non-dividend-paying stock with a strike price of $50 when the current stock price is $50, the risk-free interest rate is 10% per annum,...

Study smarter with the SolutionInn App


