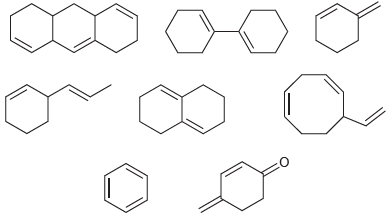
Circle each compound that has a conjugated Ï system:
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)

Answered By

Daniel Kimutai
I am a competent academic expert who delivers excellent writing content from various subjects that pertain to academics. It includes Electronics engineering, History, Economics, Government, Management, IT, Religion, English, Psychology, Sociology, among others. By using Grammarly and Turnitin tools, I make sure that the writing content is original and delivered in time. For seven years, I have worked as a freelance writer, and many scholars have achieved their career dreams through my assistance.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The infrared spectra for three compounds are provided. Each compound has one or more of the following functional groups: conjugated ketone, ester, amide, nitrile, and alkyne. Determine the functional...
-
Circle the conjugated pi bonds, if any, in the following compounds? c) CH,CH=CH-C=N b) CH3-CH=CH-C-CH3 d) H-C=C-CH=CH-C-H La e) CH-CH=CH-CH-C-CH3
-
(a) Which other compounds in Section 13.5 are conjugated dienes? (b) Which other compounds are isolated dienes? (c) Which compound is an isolated enyne?
-
State whether the following statements are true or false. Also, very briefly mention the reason if the statement is false. i. TOFO Surfactants increase the surface tension of water. ii. TOFO The Mach...
-
Estimate the values of the coefficient of earth pressure at rest, Ko, for the four soils of Problem 12-31.
-
The Open Dentistry Journal (Vol. 4, 2010) published a study on a revised tool for assessing research reports in health care. Recall that the assessment tool was validated on five systematic reviews...
-
A SIF uses two discrete inputs and one discrete output. Based on the failure rates in Table 11-2, what is the total DU failure rate for the logic solver in this safety instrumented function?
-
1. Stork Corporation (E & P of $850,000) has 1,000 shares of common stock outstanding. The shares are owned by the following individuals: Lana Johnson, 450 shares; Lori Johnson (Lanas sister), 450...
-
You are a Morgan Stanley portfolio manager of a risky portfolio with an expected rate of return of 19% and a standard deviation of 28%. The T-bill rate is 7%. Suppose your client decides to invest in...
-
The following is a list of 24 Marcos Pizza stores in Lucas County. The stores are identified by numbering them 00 through 23. Also noted is whether the store is corporate owned (C) or manager owned...
-
Draw the structure of each of the following compounds: (a) 1,4-Cyclohexadiene (b) 1,3-Cyclohexadiene (c) (Z)-1,3-Pentadiene (d) (2Z,4E)-Hepta-2,4-diene (e) 2,3-Dimethyl-1,3-butadiene
-
Firm A and Firm B are competitors in the same industry. The two firms have similar operating costs, except Firm A has more fixed operating costs than Firm B. As a result, which firm would experience...
-
What criteria are used to classify species in the Linnaean system?
-
During the financial crisis in October 2008, the federal government could borrow at a rate of 2.73% (the yield on five-year Treasury securities). During October 2008, though, Baa borrowers (corporate...
-
Xecor makes several drugs, including Anxless, approved by the FDA for the treatment of anxiety. Recent studies sponsored by Xecor indicate that Anxless may be a promising treatment for hypertension....
-
A hospital pharmacy dispensed DrugX to an inpatient pursuant to a medication order. A MedGuide is available for DrugX. When the nurse administered the drug to the patient, the patient asked if there...
-
What were the reasons that originated the real estate bubble of the early to mid-2000s? Do some research to justify your reasoning.
-
Even though this case is not an FDCA case and is really about damages, it highlights important issues under the FDCA, including substitution without authorization and the issue of generic and...
-
Access the Internet and go to www.irs.gov and select Search Forms & Instructions. Enter 4868 into the search box. What is Line 6 of the Form 4868? a. Total payments b. Balance due c. Your name d....
-
Which of the followingcarbocations is the least stable? CH3CH2 . CH3CHCH3 CH3 I . CH3C0 T CH3 IV. V. CH3 CH3CCH2 CH3
-
Answer Problem 21.37 (or reaction of the listed reagents with Propanamide.
-
What product would you expect to obtain from Grignard reaction of an excess of phenyl magnesium bromide with dimethyl carbonate, CH 3 OCO 2 CH 3 ?
-
Treatment of 5-aminopentanoic add with DCC (dicyclohexylcarbodiimide)) yields a lactam. Show the structure of the product and the mechanism of the reaction.
-
Melissa Cutt is thinking about buying some shares of EZLawn Equipment, at $36.44 per share. She expects the price of the stock to rise to $43.62 over the next 3 years. During that time she also...
-
At the beginning of the week, Taylan's restaurant had a beginning inventory of food and beverages totaling $4,000. During the week, additional food and beverages purchases totaled $3,000. At the end...
-
1. Identify the auditor's role in the internal and external auditing processes. 2. Identify one law, regulation, and statue affecting healthcare

Study smarter with the SolutionInn App


