Compare the following two constitutional isomers. The 13 C NMR spectrum of the first compound exhibits five
Question:
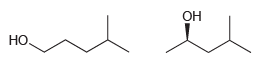
Transcribed Image Text:
Он НО.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
The first compound lacks a chirality center The two methyl groups are enantiot...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw two constitutional isomers of cis-1, 2-dibromo-cyclopentane.
-
Write structural formulas of the type indicated: (a) Bond-line formulas for seven constitutional isomers with the formula C4H10O; (b) Condensed structural formulas for two constitutional isomers with...
-
Two constitutional isomers having molecular formula C4H6O are both symmetrical in structure. In their infrared spectra, neither isomer when in dilute solution in CCl4 (used because it is nonpolar)...
-
An employee of Gus's Restaurant sustains a work-related injury. The owner of the restaurant is liable for the injury, however, the employee is not covered under the state's workers' compensation...
-
What are the differences between a managerial and a technical designer of a DSS?
-
A linear model fit to predict weekly Sales of frozen pizza (in pounds) from the average Price ($/unit) charged by a sample of stores in the city of Dallas in 39 recent weeks is: Saks = 141,865.53 -...
-
With a neat sketch describe vapour compression refrigeration system.
-
Perform the following estimations without using ca calculator. (a) Estimate the mass of water (kg) in an Olympic-size swimming pool. (b) A drinking glass is being filled from a pitcher. Estimate the...
-
Consider the soil profile shown in Figure E3-2. The moist unit weight of the top sand layer is 20.0 kN/m. Capillary rise is present above the groundwater table. The saturated unit weights of clay 1...
-
Figure 3.26 shows the first five peaks of the x-ray diffraction pattern for tungsten (W), which has a BCC crystal structure; monochromatic x-radiation having a wavelength of 0.1542 nm was used. (a)...
-
Explain why the effective interest rate (r EAR ) on a loan often is much higher than the simple, or stated, interest rate (APR).
-
Susan Visscher, owner of Visschers Hardware, is negotiating with First Merchants Bank for a $50,000, one-year loan. First Merchants has offered Visscher the following alternatives. Calculate the...
-
Define the terms control and manipulation. Describe a possible Lab experiment where you would need to control a variable. Further, include a possible variable over which you would have no control,...
-
To what extent is the new psychological contract a myth dreamed up by HR commentators to add a new dimension to discussions about SHRM?
-
Analyse the significance in the growth of multi-national companies;
-
Define strategic international human resource management;
-
In January 2019, Judd Harrison decides to sell 100 shares in the Widget Company. Since April 2018, no stock dividends had been paid by the company. On the date the stock is sold the market price is...
-
Identify the key components of strategic international human resource management;
-
A. Bea Jones (birthdate March 27, 1984) moved from Texas to Florida in December 2017. She lives at 654 Ocean Way, Gulfport, FL 33707. Beas Social Security number is 466-78-7359 and she is single. Her...
-
(a) Bright Sdn Bhd (BSB) is a tax resident manufacturing company in Johor, which involves in ceramic tiles. Currently, BSBs annual sales turnover has been forecasted to be around RM 300,000 for the...
-
The amino acid methionine is biosynthesized by a multistep route that includes reaction of an imine of pyridoxal phosphate (PLP) to give an unsaturated imine, which then reacts with cysteine. What...
-
Each of the following reaction schemes contains one or more flaws. What is wrong in each case? How would you correct eachscheme? (a) Ag*, NH,OH 1. CH3MgBr 2. * CH , I b) H H2 CH * CHgCHHIO)2 (c) ...
-
6-Methyl-5-hepten-2-one is a constituent of lemongrass oil. How could you synthesize this substance from methyl4-oxopentanoate? CH3CH2CH2OCH3 Methyl 4-oxopentanoate
-
The wheels of a skateboard roll without slipping as it accelerates at 0.45 m/s down an 80-m-long hill. Part A If the skateboarder travels at 1.5 m/s at the top of the hill, what is the average...
-
A 2.30 g air-inflated balloon is given an excess negative charge, q = -3.50 10-8 C, by rubbing it with a blanket. It is found that a charged rod can be held above the balloon at a distance of d =...
-
A positive charge q = 2.60 C on a frictionless horizontal surface is attached to a spring of force constant k as in the figure shown below. When a charge of 42 = -8.350 C is placed 9.50 cm away from...

Study smarter with the SolutionInn App


