Consider the following reaction: (a) Use Table 6.1 to estimate ÎH for this reaction. (b) ÎS of
Question:
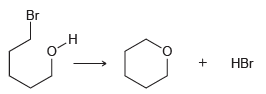
(a) Use Table 6.1 to estimate ΔH for this reaction.
(b) ΔS of this reaction is positive. Explain.
(c) Determine the sign of ΔG.
(d) Is the sign of ΔG dependent on temperature?
(e) Is the magnitude of ΔG dependent on temperature?
Transcribed Image Text:
Br o-н HBr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a Bonds Broken kJmol Bonds Formed kJmol RCH 2 x Br ...View the full answer

Answered By

Rajat Gupta
used to take tution classes from my school time.
Conducted special topic claases during my graduation to help the students pass their exams.
Currently, teaching and conducting online claases during my post- graduation too.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following reaction at some temperature: H2O(g) + CO(g) H2(g) + CO2(g) K = 2.0 Some molecules of H2O and CO are placed in a 1.0- L container as shown below. When equilibrium is reached,...
-
Consider the following reaction between oxides of nitrogen: NO2(g) + N2O(g) 3 NO (g) (a) Use data in Appendix C to predict how G for the reaction varies with increasing temperature. (b) Calculate G...
-
Consider the following reaction at 298 K: 2H2(g) + O2(g) 2H2O(l) H = 2571.6 kJ/mol Calculate Ssys, Ssurr, and Suniv for the reaction.
-
Evaluate the limit of the sequence or state that it does not exist. an || u8 n!
-
Suppose that you are sole proprietor presenting to a group of investors where you are seeking 20 million dollars to raise capital for your manufacturing company. Choose the one form of organization...
-
Use the definition of expected value for a discrete random variable to compute expectations, given a \(p d f f(x)\) and a function \(g(X)\) of \(X\).
-
The pure shrinkage estimator is defined as \(\hat{\beta}_{s}=c \hat{\beta}\), were \(0 \leq c \leq 1\) is a constant chosen by the analyst. Describe the kind of shrinkage that this estimator...
-
Information concerning Montana Company is provided in BE19-16. What are the total product costs for the company under absorption costing?
-
LIFO Liquidation Profit Hammond Company adopted LIFO when it was formed on January 1, 2017. Since then, the company has had the following purchases and sales of its single inventory item: Year Units...
-
You, CPA, are working as the controller for a video game development company called All Starr Games Inc. (All Starr). The company develops sports-related games, and its recent virtual rugby game was...
-
In each of the following cases compare the bonds identified with red arrows, and determine which bond you would expect to have the largest bond dissociation energy: a. b. CI .F Br
-
Consider the following four energy diagrams: (a) Which diagrams correspond with a two-step mechanism? (b) Which diagrams correspond with a one-step mechanism? (c) Compare energy diagrams A and C....
-
One of the more interesting magazines to review about current trends in American culture is American Demographics magazine. Go to the Web site of this magazine at...
-
Jennifer Yauger owned a loft in uptown Silverton. Robert Tomba and Gary Jorgen were partners in a mail-order business in Middletown called NASCAR Collectibles. To open an outlet in Silverton, Tomba...
-
Cramers mortgage contained a provision requiring her to pay monthly tax and insurance payments into an escrow account held by the bank in addition to principal and interest. Cramer paid the principal...
-
An economy is in a recession with a large recessionary gap and a government budget deficit. a. Is the government budget deficit a structural deficit or a cyclical deficit? Explain. b. Explain how...
-
Would business investment be encouraged or discouraged between the time of the announcement and the actual increase in the interest rate? Why or why not? To combat high and rising inflation, the Bank...
-
Why might Venezuelas monetary authorities decide to cut interest rates in this situation? To work Problems 28 to 30, use the information that Venezuelas economy is expected to witness a staggering...
-
What physics principle does Manuel use when he pumps in rhythm with the natural frequency of the swing?
-
Discuss the information available from the following techniques in the analysis of inorganic pigments used in antique oil paintings: (i) Powder X-ray diffraction, (ii) Infrared and Raman...
-
What product would you expect to obtain from the base-catalyzed Michael reaction of (a) 1, 3-diphenylprop-2-en-1-one (Section 19.5A) and acetophenone (b) 1, 3-diphenylprop-2-en-1-one and...
-
Since the products obtained from Claisen condensations are b-keto esters, subsequent hydrolysis and decarboxylation of these products give a general method for the synthesis of ketones. Show how you...
-
When acrolein (propenal) reacts with hydrazine, the product is a dihydropyrazole: Suggest a mechanism that explains this reaction. H + H2N-NH2 Acrolein Hydrazine A dihydropyrazole
-
How do prokaryotes engage in sophisticated social behaviors, such as altruism and kin selection, and what genetic and ecological factors influence the evolution of these traits?
-
N Suppose for the integral T|[a,b] () = f f(x)dx, we approximate it with the midpoint method, which for mesh {x;} where xj = a + , = b-a N j=0 is given by N-1 1 A[a,b] (f) = 5x f (xj+1/2), where...
-
What are the structural and functional complexities of prokaryotic cell membranes, and how do they contribute to processes such as nutrient uptake, osmoregulation, and signal transduction?

Study smarter with the SolutionInn App


