Consider the structure of beta-carotene, mentioned earlier in this chapter: Design a synthesis of beta carotene using
Question:
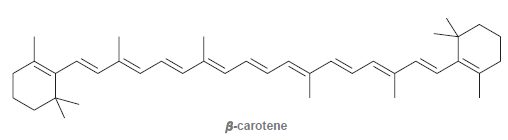
Design a synthesis of beta carotene using the compound below as your only source of carbon atoms:
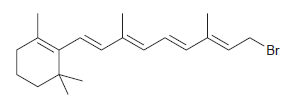
Transcribed Image Text:
B-carotene Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
1 PPh3 2 B...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What organic compound mentioned earlier in this chapter is a cyclic dimer of OCH2CH2 units
-
What organic compound mentioned earlier in this chapter is a cyclic dimer of OCH2CH2 units?
-
Using acetylene as your only source of carbon atoms, outline a synthesis for 3-hexyne.
-
How do you ensure data quality?
-
In late 2013 and early 2014, Target Corporation reported a data breach possibly affecting more than 100 million customer credit and debit card accounts. Plaintiffs brought a shareholder derivative...
-
Construct a 99% confidence interval estimate for the difference in the number of exercises professors think are necessary and the number the students deem necessary for the situation described in...
-
According to the Congressional Research Service, 2,191,011 people are employed by the federal government in the United States. Suppose that a random sample of 3,500 of these federal employees was...
-
Alternative methods of joint-cost allocation, product-mix decisions. The Sunshine Oil Company buys crude vegetable oil. Refining this oil results in four products at the splitoft point; A, B, C, and...
-
We use demand elasticities to quantify how much quantity demanded for a product would change given changes to the factors that affect demand. Some common demand elasticities are: own-price...
-
Discuss the mechanism of attribute/relationship inheritance. Why is it useful?
-
Identify the reagents necessary to prepare each of the following compounds using a Wittig reaction: (a) (b) (c) (d) (e) -
-
Identify the reagents necessary to accomplish each of the transformations below: (a) (b)
-
How did Ralph Lauren violate the FCPA? Why did Ralph Lauren receive less severe penalties for the misconduct? How can the Ralph Lauren bribery case set a precedent for other firms when discovering...
-
Give the reasons that banks select the bank holding company form of organization. Why would a bank holding company seek to convert to a fi nancial holding company?
-
Look at the MOW research on what people value about work and what needs their jobs satisfied. Do you think that working from home will be added as an insight, post COVID-19?
-
JAMS Technologies, Inc., owed $77,000 to Easy Luck Company in Miami, Florida. To pay the debt, JAMS presented a check drawn on the account of Lanco Manufacturing Corporation in Banco Bilbao Vizcaya...
-
The boiling point for species A at 1 bar is reported to be 250 K, and you desire to know the boiling point at 10 bar. Knowing the enthalpy of vaporization, you apply the Clausius-Clapeyron equation...
-
Choose a country that scores very differently than your own on Hofstedes cultural dimensions of power distance, masculinity, and individualism. How would these differences require a different...
-
What is the smallest (most negative) 32-bit binary number that can be represented with (a) Unsigned numbers? (b) Twos complement numbers? (c) Sign/magnitude numbers?
-
On January 1, 2018, Khalid Ltd., which follows IAS 17, entered into an eight-year lease agreement for three dryers. Annual lease payments for the equipment are $28,500 at the beginning of each lease...
-
Give IUPAC names for the following alkenes: (a) (b) (c) (d) (e) (f) OH CI
-
Write bond-line formulas for the following: (a) cis-3-Octene (b) trans-2-Hexene (c) 2,4-Dimethyl-2-pentene (d) trans-1-Chlorobut-2-ene (e) 4,5-Dibromo-1-pentene (f)...
-
Give the structures and IUPAC names for all the alkynes with the formula C6H10.
-
J is going to receive a 30-year annuity of 8,500 and L is going to receive perpetuity of 8,500. If the appropriate interest rate is 6%, how much more is L's cash flow worth?
-
. Assume that over the past 88 years, U.S. Treasury bills had an average return of 3.5% as compared to 6.1% on long-term government bonds. What was the average risk premium on the long-term...
-
The following data were gathered to use in reconciling the bank account of Bradford Company: Balance per bank $ 18,050 Balance per company records 10,040 Bank service charges 50 Deposit in transit...

Study smarter with the SolutionInn App


