Consider the two methyl groups shown in the following compound. Explain why the methyl group on the
Question:
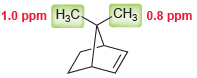
Transcribed Image Text:
CH3 0.8 ppm 1.0 ppm H;C.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
The methyl group on the righ...View the full answer

Answered By

Collins Omondi
I have been an academic and content writer for at least 6 years, working on different academic fields including accounting, political science, technology, law, and nursing in addition to those earlier listed under my education background.
I have a Bachelor’s degree in Commerce (Accounting option), and vast knowledge in various academic fields Finance, Economics, Marketing, Management, Social Science, Women and Gender, Business law, and Statistics among others.
4.80+
4+ Reviews
16+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The 1H NMR spectrum of N, N-dimethylformamide shows a separate signal for each of the two methyl groups. Can you explain why?
-
Approximately how much steric strain does the 1, 3-diaxial interaction between the two methyl groups introduce into the diaxial conformation of cis-1, 3-dirnethylcyclohexanc?
-
3-Methyl-2-butanol has five signals in its 13C NMR spectrum at 17.90, 18.15, 20.00, 35.05, and 72.75 ?. Why are the two methyl groups attached to C3 nonequivalent? Making a molecular model should be...
-
Discuss how efficiency and effectiveness are used to aid in decision making within a healthcareorganization. Is one more important than the other? Describe the importance of financial information in...
-
1. How does Pandora Radio recommend music to its listeners? 2. How are listeners able to create their own customized stations? 3. What are some variables that Pandora Radio uses to recommend a song?
-
Fill in the blanks to make the following statements correct. a. On a graph with Y on the vertical axis and X on the horizontal axis, the slope of a straight line is calculated as ___________. b. In...
-
What considerations are to be taken into account for the balancing of a reciprocating engine?
-
Two or more items are omitted in each of the following tabulations of income statement data. Fill in the amounts that aremissing. 2006 2007 2008 $290,000 $ $410,000 Sales Sales returns Net sales...
-
what ways do mindfulness-based stress reduction (MBSR) techniques contribute to reducing stress, and what are their limitations ?
-
Write a program that will take as input two Web page URLs and find a path of links from one to the other. What is an appropriate search strategy is bidirectional search a good idea? Could a search...
-
Indicate how each of the following might affect the data shown in the table and graph in Figure 2 of this appendix: a. GSUs athletic director schedules higher-quality opponents. b. An NBA team...
-
The accompanying graph shows curve XX and tangents at points A , B , and C . Calculate the slope of the curve at these three points.
-
Contrast the five bases of power?
-
RECEPTIONIST DEALERSHIP Briefly describe the task(s) The receptionist at a car dealership Working at a car dealership has shown me several benefits, including: Sales experience, I have gained...
-
PetValue, Inc. has 100,000 shares outstanding and seven directors. 1. What number constitutes a quorum of shareholders? 2. What number constitutes a quorum of directors? 3. If six directors are...
-
In December 2015, Joan Jett bought the candy store known as "Sweet Dreams" from Molly Cass. Sweet Dreams was located on 5 th Street in Sunnyville. As part of the contract, Molly Cass was not...
-
Due to the fall out of Jim's actions, ITC Classmate asks Germ Destroyer to attempt to negotiate a resolution to their differences. Germ Destroyer offers to change the name and design of its product...
-
Austin, the President of Germ Destroyer, is terrified of Jim, and signs a recission agreement. When Jim leaves his office, he immediately calls 911. The police finds him sweating and breathing...
-
Evaluate the expression. - 2:)(-2 + V25) (4 2)(-2 + V25)
-
Don Griffin worked as an accountant at a local accounting firm for five years after graduating from university. Recently, he opened his own accounting practice, which he operates as a corporation....
-
Each of the following carbocations can rearrange to a more stable ion. Propose structures for the likely rearrangement products. H, (a) CH3CH2CH2CH2* (b) CH3CHCHCH3 CH CH CH2* (c)
-
Addition of HCI to 1-isopropylcyclohexene yields a rearranged product. Propose a mechanism, showing the structures of the intermediates and using curved arrows to indicate electron flow in each step....
-
Addition of HCI to 1-isopropcnyl-1-methylcyclopcntane yields 1-chloro-1, 2, 2- trimethylcyclohexane. Propose a mechanism, showing the structures of the intermediates and using curved arrows to...
-
Resources Unlimited Corporation was formed in 1985 through the merger of two natural pipeline companies. The result was the largest gas distribution network in the United States, with 38,000 miles of...
-
Toyota was the world's first automobile manufacturer to produce more than 10 million vehicles per year, which it has done since 2012, when it also reported the production of its 200-millionth...
-
According to the video "The Importance of Budgeting," budgeting is a crucial aspect of running a successful business as it helps administrators allocate resources effectively to meet their goals. It...

Study smarter with the SolutionInn App


