Determine the hybridization state of each carbon atom in the following compounds: a. b. , C=c=c=C
Question:
a.
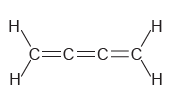
b.
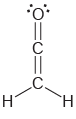
Transcribed Image Text:
Н, н C=c=c=C Н н || Н `H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)
a b ...View the full answer

Answered By

Hemstone Ouma
"Hi there! My name is Hemstone Ouma and I am a computer scientist with a strong background in hands-on experience skills such as programming, sofware development and testing to name just a few. I have a degree in computer science from Dedan Kimathi University of Technology and a Masters degree from the University of Nairobi in Business Education. I have spent the past 6 years working in the field, gaining a wide range of skills and knowledge. In my current role as a programmer, I have had the opportunity to work on a variety of projects and have developed a strong understanding of several programming languages such as python, java, C++, C# and Javascript.
In addition to my professional experience, I also have a passion for teaching and helping others to learn. I have experience as a tutor, both in a formal setting and on a one-on-one basis, and have a proven track record of helping students to succeed. I believe that with the right guidance and support, anyone can learn and excel in computer science.
I am excited to bring my skills and experience to a new opportunity and am always looking for ways to make an impact and grow as a professional. I am confident that my hands-on experience as a computer scientist and tutor make me a strong candidate for any role and I am excited to see where my career will take me next.
5.00+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the hybridization state of each carbon in the following Compounds: (b) Formaldehyde (H2C=O) (c) Ketene (H2 C=C=O) (d) Propane (CH3CH=CH2) (e) Acetone [(CH3)2C=O] (f) Acrylonitrile (CH2=CHC CPN)
-
Give the hybridization state of each carbon in the following compounds: (b) Formaldehyde (H2C==O) (c) Ketene (H2C==C==O) (d) Propene (CH3CH==CH2) (e) Acetone [(CH3)2C==O] (f) Acrylonitrile (CH2==CHCN)
-
Below are the structures of two common over-the-counter pain relievers. Determine the hybridization state of each carbon atom in these compounds: a. b. TH. H H H. .C. TH. .C: .. `H Acetylsalicyclic...
-
In seawater, the pressure p is related to the depth d according to 33p - 18d = 495 where d is in feet and p is in pounds per square inch. (a) Solve this equation for p in terms of d. (b) The Titanic...
-
"You know, we recently had a soft drink product (an exotic berry seltzer line) go through one of those simulated test markets, and it was a disaster. The new products people forgot completely about...
-
What is meant by 'useful life' and how is it determined?
-
The amount of heat required to raise the temperature by \(1^{\circ}\) of \(1 \mathrm{~g}\) of a substance is known as its (a) Heat capacity (b) Thermal conductivity (c) Specific heat (d) None of...
-
Nakashima Gallery had the following petty cash transactions in February of the current year. Feb. 2 Wrote a $ 400 check, cashed it, and gave the proceeds and the petty cashbox to Chloe Addison, the...
-
what is the difference between Activity Based Costing and Traditional Costig ?
-
1. How is the SSM different from Deming's PDCA cycle? 2. Prepare a cause-and-effect or fishbone diagram for a problem such as "Why customers have long waits for coffee." Your fishbone diagram should...
-
Identify the reagents you would use to accomplish the following transformation. CI
-
At year-end 2009, total assets for Shome Inc. were $1.2 million and accounts payable were $375,000. Sales, which in 2009 were $2.5 million, are expected to increase by 25 percent in 2010. Total...
-
The following information pertains to Nell Companys production of one unit of its manufactured product during the month of June. The company recognizes the materials price variance when materials are...
-
Consider a $1,000 corporate bond with a 3.5% coupon that pays interest quarterly and matures in five years. If its yield to maturity is 4%, its price would be closest to?
-
Basic policies for uniprocessor scheduling are First-Come-First-Serve (FCFS), Round-robin, Shortest Process Next (SPN) and Shortest Remaining Time (SRT) scheduling policies. Consider the following...
-
(15 points) On June 1, Grill Master Ltd. had an inventory of 10 barbeques at a cost of $440 each. Grill Master uses a periodic inventory system. During the month of June the following transactions...
-
Youre having a first meeting with a potential customer at a company that you have been told wears jeans and t-shirts to work, even the CEO. What would be the appropriate attire for this meeting?
-
Chloe's Cafe bakes croissants that it sells to local restaurants and grocery stores. The average costs to bake the croissants are $0.30 for 3,100 and $0.25 for 6,200. Required: If the total cost...
-
Find the product. (2m 3 + n)(2m 3 - n)
-
According during to the IRS, individuals filing federal income tax returns prior to March 31 received an average refund of $1,088 in 2018. Consider the population of "last-minute" filers who mail...
-
Draw an energy diagram for a reaction with Keq = 1. What is the value of G in this reaction?
-
The addition of water to ethylene to yield ethanol has the following thermodynamic parameters: (a) Is the reaction exothermic or endothermic? (b) Is the reaction favorable (spontaneous) or...
-
When a mixture of methane and chlorine is irradiated, reaction commences immediately. When irradiation is stopped, the reaction gradually slows down but does not stop immediately. Explain.
-
Suppose Stocks A, B and C are the only three component stocks in a benchmark index. The number of shares outstanding of Stocks A, B and C are 369,000 shares, 327,000 shares, and 240,000 shares,...
-
Botanists have determined that some species of weed grow in a circular pattern. For one such species, the area A , in square meters, can be approximated by A ( t ) = 0.004 t 2 , where t is the time...
-
Book Print ciences Gallatin Carpet Cleaning is a small, family-owned business operating out of Bozeman, Montana. For its services, the company has always charged a flat fee per hundred square feet of...

Study smarter with the SolutionInn App


