Draw a resonance structure of the compound below, which was isolated from the fruits of Ocotea corymbosa,
Question:
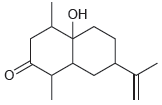
Transcribed Image Text:
Он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
H...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw a resonance structure of the compound shown below, called 2-heptanone, which is found in some kinds of cheese.
-
Draw a resonance structure of the acetonitrile anion, :CH 2 C N, and account for the acidity of nitriles.
-
Consider the structure of the compound below. (a) Identify each position where an S N 2 reaction is likely to occur. (b) Identify each position where an S N 1 reaction is likely to occur. Cl CI TsO....
-
The P04_18.xlsx file contains a single-table Data Model on a companys sales and a single blank worksheet. The goal is to create a pivot table that shows the ratio of average revenue per transaction...
-
Wanda Collier worked for Turner Industries Group, LLC, in the maintenance department. She complained to her supervisor that Jack Daniell, the head of the department, treated her unfairly. Her...
-
At the end of the financial year, prepare the general journal to close out the accounts. Post the journal to the T accounts shown in figure 9.18. Discount Received Balance Commission Received Balance...
-
What is the scope of the discovery?
-
The trial balance for Mackle Fitness, shown below, does not balance. The following errors were detected: a. The cash balance is overstated by $6,000. b. Rent expense of $3,000 was posted as a credit...
-
Let San be an infinite series such that Sy=3-- n=1 10 16 (a) What are the values of a, and an? 2.94 n=1 n=1 16 n=4 = (b) What is the value of a3? a3 = (c) Find a general formula for an an= (d) Find...
-
Assume that we make an enhancement to a computer that improves some mode of execution by a factor of 10. Enhanced mode is used 50% of the time, measured as a percentage of the execution time when the...
-
Consider a gas mixture in a 1.50-dm 3 flask at 22.0C. For each of the following mixtures, calculate the partial pressure of each gas, the total pressure, and the composition of the mixture in mole...
-
A mixture of H 2 and NH 3 has a volume of 139.0 cm 3 at 0.00C and 1 atm. The mixture is cooled to the temperature of liquid nitrogen, at which ammonia freezes out and the remaining gas is removed...
-
Refer to CVS's annual report in the Supplement to Chapter 1 and to the following data (in millions) for Walgreens: cost of goods sold, $51,692 and $48,444 for 2011 and 2010, respectively;...
-
Repeat problem 9.b to produce a goal-driven solution. Data from problem 9.b b. Generate the state space and stages of working memory for the data-driven solution to the example in Chapter 3.
-
Solve the problem using direct substitution method Maximize $f=3 x_{1}^{2}+2 x_{2}^{2}$ subject to the constraints \[x_{1}+3 x_{2}=5\]
-
When fractional factorial design is useful?
-
What is the difference between an initial value problem and a final value problem?
-
What is meant by expected value of any random variable?
-
Explain why it is important to trace transfers between bank accounts on either side of the end of the reporting period.
-
An auto-parts manufacturer is considering establishing an engineering computing center. This center will be equipped with three engineering workstations each of which would cost $25,000 and have a...
-
Write stereochemical formulas for all of the products that you would expect from each of the following reactions. (You may find models helpful.) (a) (b) (c) (d) (1) OsO4 (2) NaHSO3, H2O (1) OsO (2)...
-
(a) Write a mechanism for the following reaction. (b) What general conditions would you use to ensure a good yield of the product? (c) What general conditions would you use to carry out the reverse...
-
Give (R, S) designations for each different compound given as an answer to Problem 8.39. In Problem 8.39 (a) (b) (c) (d) (1) OsO4 (2) NaHSO3, H2O (1) OsO (2) NaHSO, HO Br Br2
-
7. (30 pts) A particle confined to the surface of a sphere is in the state 2 where (N =-0).0 <0
-
According to your book, the US CDC reported that in 2011-2012 31.5% of US adults over 20 years old were obese and 69% were obese or overweight. What was the percentages in 1950?
-
firm a and firm b are both all-equity firms. Firm A has 2 million shares outstanding trading at $68 erach. Firm B has 1.25 million shares outstanding trading at $42 each. Firm A would like to acquire...

Study smarter with the SolutionInn App


