Draw bond-line structures for vitamin A and vitamin C: . 1 -
Question:
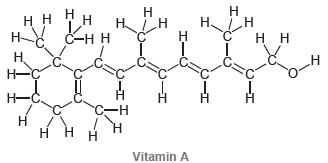
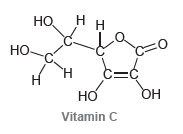
Н Н. н Н н 1н нн н С-нн Нн нн Н Н ннн Н Vitamin A но. нн Но. C=Ć он н но Vitamin C
Step by Step Answer:
Vitami...View the full answer



Related Video
Lemon juice preserves apples by slowing down the oxidation process. Oxidation is a chemical reaction that occurs when oxygen reacts with certain substances, such as apples. When an apple is cut or bitten, oxygen is exposed to the inside of the apple and causes enzymes in the apple to turn brown, which is an indication of oxidation. The browning process is caused by the production of polyphenol oxidase (PPO) enzymes that convert phenolic compounds into quinones, which then polymerize to form the brown pigments. One of the compounds present in lemon juice is ascorbic acid (vitamin C), which is a natural antioxidant. Antioxidants work by neutralizing the free radicals that cause oxidation. When lemon juice is applied to apples, the ascorbic acid in the lemon juice reacts with the PPO enzymes and slows down the browning process. You can do an experiment by cutting apples into small pieces, leaving one apple piece in contact with air and the others covered with lemon juice and compare the browning process. This will help to understand the antioxidation process in fruits.
Students also viewed these Sciences questions
-
Draw bond-line structures for all constitutional isomers with molecular formula C 4 H 9 Cl.
-
Draw bond-line structures for all constitutional isomers of the following compound: CH 3 CH 2 CH(CH 3 ) 2
-
Draw bond-line structures for all constitutional isomers of C 4 H 10 ?
-
Darrel & Co. makes electronic components. Chris Darrel, the president, recently instructed Vice President Jim Bruegger to develop a total quality control program. If we dont at least match the...
-
On March 1, Jordan Company borrows $150,000 from Ottawa State Bank by signing a 6-month, 8%, interest-bearing note. Instructions prepare the necessary entries below associated with the note payable...
-
This table gives further details of the multiple regression estimated in Exercise 32. Assume that the MRM satisfies the conditions for using this model for inference. (a) Fill in the column of...
-
What mix of inputs should be used to produce a given level of output?
-
Gomez Company issued $380,000, 7%, 10-year bonds on January 1, 2014, for $407,968. This price resulted in an effective-interest rate of 6% on the bonds. Interest is payable annually on January 1....
-
If an investor invests in a bond that has $1,000 par and pays 7% coupons semi-annually, which matures in 3 years. Assume yield stays at 3% compound semiannually and all received coupon payments will...
-
Consider the following 0x86 program: .data array DWORD 1,2,3,4,5,6,7,8,9 aravSize -(S-array)/4 ; array .code main PROC mov ecx,arraySize-1 mov esi.OFFSET array L1: mov eax. [esi] sda mov bx,2 idiv bx...
-
Draw bond-line structures for all constitutional isomers of C 5 H 12 ?
-
How many lone pairs are found in the structure of vitamin C?
-
Why is free energy free?
-
If a company sells more units than it produces, would the profits reported by absorption costing tend to be higher or lower than the profits reported by variable costing? Why?
-
Explain the significance and objective of a break-even chart and state the factors which would cause the break-even point to change.
-
Direct costing produces income statements that are a more accurate reflection of the true profit fluctuations than the income statements produced by absorption costing. State briefly why you either...
-
Define break-even analysis and outline its uses and applications.
-
A break-even chart must be used with intelligent discrimination, with an adequate grasp of assumptions underlying the technique surrounding its practical application. Elucidate the statement giving...
-
What is the confidence for fins, given that a tank has been purchased?
-
Find the volume of the described solid S. A frustum of a right circular cone with height h, lower base radius R, and top radius r -r- --R
-
Compounds A, B, and C are isomers with the formula C5H11Br. Their broadband proton-decoupled 13C NMR spectra are given in Fig. 9.32. Information from the DEPT 13C NMR spectra is given near each peak....
-
In contrast to 2-methylbutane and neopentane, the mass spectrum of 3-methylpentane (not given) has a peak of very low relative abundance at - 15. It has a peak of very high relative abundance at -...
-
Propose structures and fragmentation mechanisms corresponding to ions with m/z 57 and 41 in the mass spectrum of 4-methyl-1-hexene. onization (loss of a electron) mlz 57 m/z 41
-
Large-scale industrial production - accompanied by massive technological change, expanding international communication networks, and pro-growth government policies-generated rapid economic...
-
How can this relate to individuals who have had a TBI and are currently homeless or marginally housed?
-
1. According to Marxian theory, are crises an economic process inherent to the accumulation dynamics of current capitalism? Justify your answer. 2. According to Marxian theory, what do you consider...

Study smarter with the SolutionInn App


