Draw the condensation product obtained when the following compound is heated in the presence of aqueous sodium
Question:
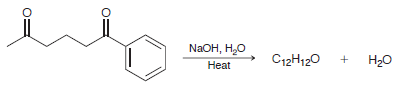
Transcribed Image Text:
NAOH, H,O Нао C12H120 Нeat
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
lilo...View the full answer

Answered By

Mary Boke
As an online tutor with over seven years of experience and a PhD in Education, I have had the opportunity to work with a wide range of students from diverse backgrounds. My experience in education has allowed me to develop a deep understanding of how students learn and the various approaches that can be used to facilitate their learning. I believe in creating a positive and inclusive learning environment that encourages students to ask questions and engage with the material. I work closely with my students to understand their individual learning styles, strengths, and challenges to tailor my approach accordingly. I also place a strong emphasis on building strong relationships with my students, which fosters trust and creates a supportive learning environment. Overall, my goal as an online tutor is to help students achieve their academic goals and develop a lifelong love of learning. I believe that education is a transformative experience that has the power to change lives, and I am committed to helping my students realize their full potential.
5.00+
4+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the condensation product obtained when each of the following compounds is heated in the presence of aqueous sodium hydroxide. (a) (b) (c) (d) (e) (f) H.
-
Draw the structure of the product obtained when the following disaccharide is treated with NaBH 4 in methanol. CH- CH- - -
-
Only a substitution product is obtained when the following compound is treated with sodium methoxide: Explain why an elimination product is not obtained. CH3 Br CH3
-
Parker Associates purchased a patent in 2018 for $200,000. The patent will be amortized over 20 years. How would Parker adjust for the annual amortization for the patent on the balance sheet? Credit...
-
What are the six categories of criminal law violations? Describe each, and rank the categories in terms of seriousness.
-
A 300 k m , 500 k V , 60 H z 300 k m , 500 k V , 60 H z three-phase uncompensated line has a positive-sequence series reactance x = 0.34 / k m x = 0.34 / k m and a positive-sequence shunt...
-
Using two beam elements, find the natural frequencies and mode shapes of the uniform fixedfixed beam shown in Fig. 12.31. p. A, I, E L 2 L 2 FIGURE 12.31 Fixed-fixed beam.
-
The balance sheet for Kronos Inc. at the end of the current fiscal year indicated the following: Bonds payable, 10% (issued in 2002, due in 2022) ...........$3,750,000 Preferred $4 stock, $40 par...
-
: In Ohio there are two manufacturers of sulfuric acid: Chemtrade and Marsulex. The marginal cost of Chemtrade is given by MCC = 2Qc and the marginal cost curve of Marsuler is represented by MCM =...
-
A piston-cylinder device contains 50 kg of water at 250 kPa and 25oC. The cross-sectional area of the piston is 0.1 m2. Heat is now transferred to the water, causing part of it to evaporate and...
-
A(n) _____ sample is obtained by dividing the population into groups and selecting all individuals from within a random sample of the groups?
-
A(n) _____ sample is obtained by dividing the population into homogeneous groups and randomly selecting individuals from each group?
-
Find a formula for the nth partial sum of the series and use it to determine if the series converges or diverges. If a series converges, find its sum. z(I + u) (-)3
-
Speaking to Pregnant Women About Prenatal Environmental Influences You have been asked by a local health department to speak to a group of newly pregnant women about prenatal environmental...
-
Felix likes people and he likes to assist families who are going through hard times. He decides he is interested in helping convicts and their families, but he soon realizes that he is uncomfortable...
-
Nigel has a contract with Susan to build garden sheds this summer. Unfortunately there has been an unforeseen catastrophic event (Hurricane), that has destroyed Nigel's business supplies which has...
-
The Jolly Scholar Brewing Company will pay a dividend of $2.50 next year.Industry analysts expect for the Company to increase its dividend at an annual rate of 7.00% in the future. You desire to earn...
-
Suppose for a one year project all outcome between a loss of 50 million and a gain of 50 million are equally likely. What is the VaR at a 95% confidence level?
-
Evaluate the following integrals. x2 8x + 16 dx (9 + 8r x)/2
-
The diagram shows the two forces acting on a small object. Which of the following is the resultant force on the object? A. 8 N downwards B. 8 N upwards C. 2 N downwards D. 2 N upwards 3 N 5 N
-
Show how 2-cyclopentyl-N,N-dimethylethanamine could be synthesized from each of the following starting materials: (a) (b) CH2 CN O- CH2 CHO (two ways)
-
Illustrate the Brgnsted basicity of Mescalino n.q by giving the structures of their conjugate acids,
-
Give the principal organic product(s) expected when N-methylaniline reacts with each of the following reagents. (a) Br2 (b) Benzoyl chloride (c) Benzyl chloride (excess), then dilute -OH (d)...
-
Write a java program to perform swapping of two numbers?
-
Consider the general case of a block of mass MB hanging at rest and a dart of mass mo moving horizontally with speed vo. The dart hits and embeds in the block, and the center of mass of the...
-
3 kg of water, originally at 10 degrees Celsius, is mixed with 10 kg of lead. The final result at equilibrium is 16 degrees Celsius. Find the initial temperature of the lead. C = 4186 kg-C (specific...

Study smarter with the SolutionInn App


