Draw the mechanism of the following reaction. This reaction is the reverse of sulfonation, so you should
Question:
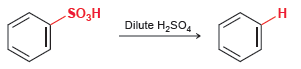
Transcribed Image Text:
SO,H .H Dilute H,SO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
1400 1 ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the mechanism of the following reaction, and make sure to draw all three resonance structures of the sigma complex. NO, HNO3 H,SO,
-
Draw the mechanism of the following reaction, which involves two consecutive Friedel-Crafts alkylations. When drawing the mechanism, do not try to draw the two alkylations as occurring simultaneously...
-
The mechanism of the following transformation involves a carbocation intermediate that rearranges in a way that we have not yet seen. Rather than occurring via a methyl shift or a hydride shift, a...
-
What line of code can you add to disable all logging messages in your program?
-
Describe some of the incidental costs of carrying and maintaining inventory for the small business owner.
-
Compare the rightmost terms of Eqs. 9.14 and 9.16 to determine whether, in any two-particle system subject to a single, constant external force, \(\Delta E\) is larger than, equal to, or smaller than...
-
Identify the control procedures that offer reasonable assurance that cash receipts are deposited intact daily.
-
Price, Inc., bottles and distributes mineral water from the companys natural springs in northern Oregon. Price markets two products: 12-ounce disposable plastic bottles and 1-gallon reusable plastic...
-
What are the differences between a finance lease and an operating lease? Why is the cost of replacement tires being ignored in the lease v buy analysis? Tori's textbook states that when making a...
-
The following are data regarding last years production of Dicer Ricer, one of the major products of Kitchen Gadget Company. During the year, 61,000 units of this product were manufactured and 62,100...
-
If the process needed to make 18 units per day, what should you do?
-
Calculate A o R and G o R for the reaction C 6 H 6 (l) + 15/2O 2 (g) 6CO 2 (g) + 3H 2 O(l) at 298 K from the combustion enthalpy of benzene and the entropies of the reactants and products.
-
Recording Transactions Including Adjusting and Closing Entries (Nonquantitative) The following accounts are used by Britts Knits, Inc. Required: For each of the following nine independent situations,...
-
As you can see by clicking on the above link, the going yield-to-maturity on a 10 year US Treasury bond is about 4.33% (as of Monday morning). Does this mean that an investor who buys this bond today...
-
The following two tables give information on treasury prices and the holdings in a portfolio managed by Bob Smith Term Structure Maturity Type Par Coupon Rate Treasury Price B spread on Treasury...
-
A 3-year term insurance policy on (50) provides a benefit of 100,000 upon death or a benefit of 50,000 upon diagnosis of disability. The policy will only pay one benefit. The benefit in either case...
-
Mountain Valley Enterprises is expected to have next year's free cash flow of $14 million. FCF is expected to grow at 5% per year into foreseeable future. Mountain Valley's cost of equity capital is...
-
S=105; X=100; rc=.02; T=60 days; standard deviation of daily returns = .012; Assume 365 calendar days in a year and 255 trading days in a year. Assume N(d1) =0.76and N(d2) = 0.74 (irrespective of...
-
Use cylindrical or spherical coordinates, whichever seems more appropriate. where E lies above the paraboloid z = x 2 + y 2 and below the plane z = 2y. Use either the Table of Integrals (on Reference...
-
The first national bank pays a 4% interest rate compound continuously. The effective annual rate paid by the bank is __________. a. 4.16% b. 4.20% c. 4.08% d. 4.12%
-
Explain why 1-chlorobicyclol2.2.llheptane, even though it is a tertiary alkyl halide, is virtually unreactive in the S"1 reaction. (It has been estimated that it is 10-13 times as reactive as...
-
Explain each of the following observations. If lithium fluoride is substituted for potassium fluoride, no reaction occurs even in the presence of the crown ether.
-
(a) Two isomeric S"2 products are possible when sodium thiosulfate is allowed to react with one equivalent of methyl iodide in methanol solution. Give the structures of the two oroducts. ...
-
Superman Enterprises has just completed an initial public offering. The firm sold 4,700,000 new shares at an offer price of $17.00 per share. The underwritering spread was $0.78 a share. The firm...
-
ABV is a non-dividend paying stock whose current price is $50. Its volatility is 12%. Over each of the next two 6-month periods the stock price is expected to go up by 9% or down by 8%. The risk-free...
-
Assume you have a portfolio consisting of a $400,000 investment in stock A and a $600,000 investment in stock B. Suppose that the daily volatilities of these two assets are 2% and 1.5%, respectively,...

Study smarter with the SolutionInn App


