Draw the monomer(s) required to make each of the following condensation polymers: (a) (b) Z Z Z
Question:
(a)
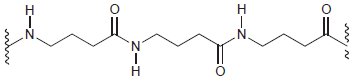
(b)
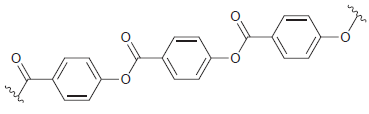
Transcribed Image Text:
エーZ Zーエ エーZ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
a ...View the full answer

Answered By

Sandeep bharal
I am a CA professional rendering full time tutoring service in the subjects of Accountancy and Finance. It is been now 10 years and i also associated with the various online platforms that help students in their assignment. I also wrote numerous of books on accountancy and finance.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the monomers required to make each of the following condensation polymers. (a) (b)
-
For each polymer shown below, (i) Draw the monomer or monomers that were needed to make the polymer. (ii) Explain whether the polymer is an addition polymer or condensation polymer. (iii) Suggest...
-
Identify the reagents necessary to make each of the following compounds with an aldol condensation. (a) (b) (c) (d) H.
-
E-max sold Dishwashers on credit, worth OMR 55,000 to customers, out of these Dishwashers customers return damaged Dishwashers for OMR 6,000. Record the entry for the Dishwashers returned? a. Debit...
-
The T-account showing the manufacturing overhead activity for Aliyah Corp. for 2018 is as follows: Requirements 1. What is the actual manufacturing overhead? 2. What is the allocated manufacturing...
-
What rules, if any, should govern mobile marketing to children?
-
To test the null hypothesis that the difference between two population proportions equals some constant \(\delta_{0}\), not necessarily 0 , we can use the statistic which, for large samples, is a...
-
The City of Phoenix, Arizona, has a variety of General Fund investments. The city had General Fund investments at July 1, 20X5, with a fair value of $29,269,000 and accrued interest receivable of...
-
Explain why the price elasticity of demand determines the incidence of a commodity tax levied on the supplier? Explain why a tax on alcohol may have a deleterious impact on the families of alcohol...
-
Carleton Company has two service departments and two production departments. Information on annual manufacturing overhead costs and cost drivers follows: The company allocates service department...
-
Determine whether the following pairs of monomers would form chain-growth polymers or step growth polymers. (a) (b) - - NO2 Ph
-
Quiana is a synthetic polymer that can be used to make fabric that mimics the texture of silk. It can be prepared from the following monomers: (a) Draw the structure of Quiana. (b) Is Quiana a...
-
What is a hypoeutectoid steel, and what structure will it assume upon slow cooling? What is a hypereutectoid steel and how will its structure differ from that of a hypoeutectoid?
-
1. Wilson Security Value demonstrated Brief Explanation of How Each Value Led to Customer Satisfaction 2. : Identify the three qualities of Wilson Security leaders. Briefly explain why each quality...
-
The Peace for All Co. paid $2,700,000 to thThe Peace for All Co. paid $2,700,000 to the Moonbeam Co. and purchased all outstanding stock of the company. Peace for All Co. assumed all of the...
-
Provide the relevant AS/NZ Standard for the following PPE items: PPE item: AS/NZ Standard: 1.Gloves 2.Eyewear 3.Gowns 4.Masks
-
On March 1, 2021, Jeff and Alex each purchased 100 common shares from corporate treasury on the incorporation of Costa Inc. for $4,000 each ($40/share) In March 2023, Jeff Bought Alex's 100 common...
-
The FIN340 Company has 920,000 shares of common stock, 150,000 shares of preferred stock, and 21,000 bonds outstanding (par value = $1,000 per bond). The current market pricing for each is...
-
Management of the Veil Acts Company just announced that instead of a regular dividend this quarter, the company will repurchase shares using the same amount of cash that would have been paid in the...
-
(a) What do data breach notification laws require? (b) Why has this caused companies to think more about security?
-
Show how you would use direct alkylation to synthesize the following compounds. (a) Benzyltrimethylammonium iodide (b) Pentan-1-amine (c) Benzylamine
-
Give the products expected from the following reactions. (a) Acetyl chloride + ethylamine (b) (c) (CHs2NIH benzoyl chloride dimethylamine NH CH3 (CH24-C -CI+ hexanoyl chloride piperidine
-
What would happen in the synthesis of sulfanilamide if the amino group were not protected as an amide in the chlorosulfonation step?
-
The highest WACC is 11%, the baseline WACC is 7.4% and lowest WACCS is 2.8%. To derive a WACC that better accounts for extreme scenarios, you decide to assign the following probability weights: High-...
-
Give 2 examples of the ff: Statutory law Regulatory law Common law Include the title of each example and explain why you chose these examples ( 1 to 2 sentences ) . Note: Write your reference ( s )
-
The Adams family includes a financially well-informed couple, both aged 36, and two children aged 4 and 6. The family is financially sound but suffered badly during the tech meltdown in 2000. The...

Study smarter with the SolutionInn App


