Draw the product formed when each of the following compounds is treated with NaNO 2 and HCl:
Question:
(a)
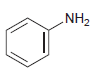
(b)
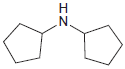
Transcribed Image Text:
NH2 N.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
a b...View the full answer

Answered By

Madhur Jain
I have 6 years of rich teaching experience in subjects like Mathematics, Accounting, and Entrance Exams preparation. With my experience, I am able to quickly adapt to the student's level of understanding and make the best use of his time.
I focus on teaching concepts along with the applications and what separates me is the connection I create with my students. I am well qualified for working on complex problems and reaching out to the solutions in minimal time. I was also awarded 'The Best Tutor Award' for 2 consecutive years in my previous job.
Hoping to get to work on some really interesting problems here.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the major product formed when each of the following compounds is treated with Et2CuLi followed by mild acid. (a) (b) (c) CN
-
Draw the enolate that is formed when each of the following compounds is treated with LDA. (a) (b) (c) (d)
-
Give the product expected when each of the following compounds is treated with acid. CO.H CO,H + heat)
-
Given the following information, prepare a production report with materials added at the beginning and ending work in process inventory being 20% complete with regard to conversion costs. Costs to...
-
A paper manufacturer buys a machine for $120,000. It depreciates at a rate of 30% per year. (In other words, at the end of each year the depreciated value is 70% of what it was at the beginning of...
-
Define Thalers concept of a financial anomaly and illustrate with some examples.
-
Test the hypotheses H0: 1 = 0 versus H1: 1 0. Use the = 0.01 level of significance. Exercises 610 refer to the following data set: x 25 13 16 19 29 19 16 30 y 40 20 33 30 50 37 34 37
-
During 2015, Walnut Company completed the following two transactions. The annual accounting period ends December 31. a. Paid and recorded wages of $130,000 during 2015; however, at the end of...
-
Tommy arranged a P10,000,000 revolving credit agreement with a group of banks. The company paid an annual commitment fee of 0.5% of the unused balance of the loan commitment. On the used portion of...
-
Bert the Payroll Guy is about to retire after 40 years and it's time to replace his manual time card system with some sort of computerized database. You have been asked to come up with the database...
-
Choose one of the following products and explain how it blurs the distinction between goods and services. a. Knee replacement surgery b. Dinner at a popular restaurant c. Purchase and installation of...
-
Consider the structure of the azo dye called alizarine yellow R (below). Show the reagents you would use to prepare this compound via an azo coupling process. .N. N' O2N
-
Cameron Levitt is the payroll accountant for Glowing Yoga. He is preparing a presentation about the Section 125 Cafeteria plans. Which of the following items may qualify for inclusion in a cafeteria...
-
For the financial data given below for the company Sheela Foams Ltd, construct the Balance Sheet, P&L and Cashflow Statements for the years 2023, 2024, and 2025. Perform the horizontal, vertical and...
-
Jillian is single and her son, Parker is 18 years old. If her AGI is $76,200, what is her child or qualifying dependent tax credit for Parker?
-
If in August Logan's sales is $3,000,000, while in July his cash collection is $2,910,000, what is Logan's total collection in August?
-
Fast Iguana had a great year. It grew its revenues by 20% while maintaining its Gross Profit Margin (GPM) at 22%. Fast Iguana reported Net Income of $95M. You are the CFO of Fast Iguana and your...
-
The owner of a pizza restaurant needs to buy a new pizza oven for the restaurant. The oven costs $500, is expected to last 5 years, and will be depreciated using the straight line method. If the...
-
The amount of monthly data usage by a sample of 10 cellphone users (in MB) was: 0.4, 2.7MB, 5.6, 4.3, 11.4, 26.8, 1.6, 1,079, 8.3, 4.2 Are there any potential irregularities in the data?
-
What are the 5 Cs of marketing channel structure?
-
Sulfur dioxide has a dipole moment of 1.60 D. Carbon dioxide has a dipole moment of zero, even though C-O bonds are more polar than S-O bonds. Explain this apparent contradiction.
-
For each of the following compounds, 1. Draw the Lewis structure. 2. Show how the bond dipole moments (and those of any nonbonding pairs of electrons) contribute to the molecular dipole moment. 3....
-
Diethyl ether and butan-1-ol are isomers, and they have similar solubilities in water. Their boiling points are very different, however. Explain why these two compounds have similar solubility...
-
The position of a particle moving along an x axis is given by x = 15.0t2 - 5.00t, where x is in meters and t is in seconds. Determine (a position, (b) the velocity, and (c) the acceleration of the...
-
Two forces act on a 2 7 . 0 kg object floating in space. One force has magnitude of 1 6 . 0 N and acts in the positive y axis direction. The second force has a magnitude of 1 0 . 2 N and acts at an...
-
You have been presented with the basic principles of personal finance, why do you think it is important to study this topic for a finance student? Among the basic principles of personal finance, what...

Study smarter with the SolutionInn App


