Draw the products of each of the following acid-base reactions, and then predict the position of equilibrium
Question:
(a)
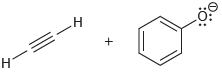
(b)
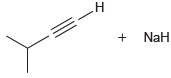
Transcribed Image Text:
н н NaH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
a...View the full answer

Answered By

Allan Olal
I have vast tutoring experience of more than 8 years and my primary objective as a tutor is to ensure that a student achieves their academic goals.
4.70+
78+ Reviews
412+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give structures for the products of each of the following reactions: (a) (b) (c) (d) (e) Nal (1 mol) H -acetone GH,H + NaBr Nal (1 mol), CH,NaCl CI CI BrBr (1 mol C4HS2 2 NaBr OH NaH (-H2) Cl Et O...
-
Give structures for the products of each of the following reactions: (a) (b) (c) (d) Product of (c) heat (e) (f) (g) Aniline + propanoyl chloride (h) Tetraethylammonium hydroxide heat (i) p-Toluidine...
-
Give the products of each of the following reactions: a. b. c. d. e. f. g. h. HCI CH CH2CH CHCH2OH excess catalytic CCH CH3 NH2NH2 CH2CH3 NH2NH 1. NaBH4 0 HCI excess CH,CH,CH,COCH CHs 2. H3o 1. LIAIH...
-
Discuss Planning & Preparation in relation to: Preparing work documents and performing document review Preparing an audit plan
-
The following information was gathered for Rogers Company for the year ended December 31, 20x4. Budgeted Actual Direct labor-hours 75,000 dlh 77,500 dlh Factory overhead $525,000 $558,000 Assume that...
-
Workers compensation is an example of which of the following types of liability? A. Absolute liability. B. Joint liability. C. Strict liability. D. Vicarious liability.
-
Team-selling situations might be a better alternative than individual selling arrangements in terms of being able to positively motivate members of the sales force. Discuss.
-
Merando Corporation was formed on January 1, 2014. At December 31, 2014, Bill Jensen, the president and sole stockholder, decided to prepare a balance sheet, which appeared as follows. Bill willingly...
-
f(x) = x In(2x) Find f(x) for the function below....
-
During the current year, the City of Plattsburgh recorded the following transactions related to its property taxes: 1. Levied property taxes of $6,600,000, of which 2 percent is estimated to be...
-
How do policy decisions affect the development of energy technologies?
-
Describe some advantages and disadvantages of the three types of fossil fuels.
-
"If nominal GDP rises, velocity must rise." Is this statement true, false, or uncertain? Explain your answer.
-
In February, 2019, the SEC announced that Deloitte Touche Tohmatsu LLC (Deloitte Japan) will pay $2 million to settle charges of independence violations for issuing audit reports for an audit client...
-
Kenyatta purchased a $5,000 face value discount note for $4,925. The note will reach maturity in 39 weeks, and the simple discount rate is 2%. What is the equivalent simple interest rate?
-
This forum serves as a platform for comprehending the intricate relationship between taxation and personal finance decisions. As you engage in discussions, consider how taxation influences your...
-
At the transport layer , to define the processes we need two identifiers _ _ _ _ _ _ _ _ _ _ _ _ .
-
where should you perform and display the results of error checking?
-
Solve each system, using the method indicated, if possible. (Gauss-Jordan) 3x + 5y = -5 -2x + 3y = 16
-
B made an issue of 150,000 $1 ordinary shares at a premium of 20% the proceeds of which is received by cheque. What is the correct journal to record this? A. Bank Share capital Share premium B. Bank...
-
N-Phenylsydnone, so-named because it was first studied at the University of Sydney Australia, behaves like a typical aromatic molecule. Explain, using the H?ckel 4n + 2 rule. :. N-Phenylsydnone
-
1-Phenyl-2-butene has an ultraviolet absorption at max = 208 nm ( = 8000). On treatment with a small amount of strong acid, isomerization occurs and a new substance with max = 250 nm ( = 15,800) is...
-
What is the structure of a hydrocarbon that has M + = 120 in its mass spectrum and has the following 1 H NMR spectrum? 7.25 (5 H, broad singlet); 2.90 (1 H, septet, J = 7 Hz); 1.22 (6 H, doublet,...
-
The Step Company has the following Information for the year just ended: Sales in units Budget 15,000 Actual 14,000 Sales $150,000 $147,000 Less: Variable Expenses 90,000 82,600 Contribution Margin $...
-
Question 2 (2 marks) The year level coordinators for Years 7 to 11 at the school are Amy (A), Brian (B), Claire (C), Daisy (D) and Ellie (E). A faulty telephone system means that some of these...
-
On a certain farm alfalfa yields have been 4.3, 7.2, 5.6, and 3.6 tons per acre in the last 4 years. What is the expected value for the farm's alfalfa yield, if each past result is given equal weight...

Study smarter with the SolutionInn App


