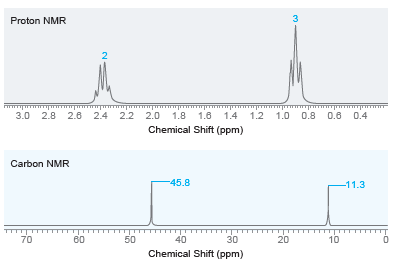
Draw the structure of the compound with molecular formula C 6 H 15 N that exhibits the
Question:
Transcribed Image Text:
3 Proton NMR 3.0 2.8 2.6 2.4 2.2 2.0 1.8 1.6 1.4 1.2 1.0 0.8 0.6 0.4 Chemical Shift (ppm) Carbon NMR -45.8 -11.3 40 10 50 70 80 30 20 Chemical Shift (ppm) 2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)

Answered By

Allan Simiyu
I am an adroit Writer. I am a dedicated writer having worked as a writer for 3 years now. With this, I am sure to ace in the field by helping students break down abstract concepts into simpler ideas.
5.00+
8+ Reviews
54+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the structure of the compound with molecular formula C8H11N that exhibits the following 1 H NMR and 13 C NMR spectra: Proton NMR 2 22 Chemical Shift (ppm) Carbon NMR 128.8 128.4 40.0 -126.1...
-
Deduce the structure of a compound with molecular formula C 6 H 14 O 2 that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. 100 80 60 40 20 3000 2500 Wavenumber (cm-1) 4000 3500 2000 1500...
-
Deduce the structure of a compound with molecular formula C 6 H 14 O 2 that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. 100 80 60 40 20 4000 3500 3000 2500 2000 1500 1000 Wavenumber...
-
Perhaps more surprising to Mr. Pitkin was a proposal by the VP of Marketing to make a major investment in market share by increasing promotional expenditures by $2.5 million during 1998-2000. Sales...
-
Without calculating, determine whether the value of nPr is greater than the value of nCr for the values of n and r given in the table. Complete the table using yes (Y) or no (N). Is the value of nPr...
-
An insurance company sells an automobile policy with a deductible of one unit. Let X be the amount of the loss having pmf Where c is a constant. Determine c and the expected value of the amount the...
-
Using the results of Problem 2.6, determine the invariants \(U_{i}\) and \(V_{i}\) for the AS/3501 lamina, where \(i=1,2,3,4\). Problem 2.6 Find all components of the stiffness and compliance...
-
Fabiano Brothers Co. is considering an investment that requires immediate payment of $550,000 and provides expected cash inflows of $100,000 annually for eight years. What is the investments payback...
-
List and Explain two different legislation relevant to detailed designs of motorways and interchanges ?
-
Consider the following premerger information about a bidding firm (Firm B) and a target firm (Firm T). Assume that both firms have no debt outstanding. Firm B has estimated that the value of the...
-
Using benzene as your only source of carbon atoms and ammonia as your only source of nitrogen atoms, propose a synthesis for the following compound: HN NH
-
Propose a mechanism for the following process: REN heat + N2 + CO2
-
A 50.0-mL sample of 0.0150 M Ag2SO4 is added to 25.0 mL of 0.0100 M PbCl2. What is the net ionic equation for the reaction that occurs? What are the concentrations of ions in the mixture at...
-
Is climate change natural or anthropogenic? Give a thorough response.
-
Why are hurricanes expected to become more frequent as the climate warms?
-
What is the IPCC? By how much does the IPCC predict climate will change over the next century?
-
The use of fossil fuels contributes to global climate change. Are there other reasons to reduce our consumption of fossil fuels? If so, describe them.
-
Name one preventable natural disaster. Name one nonpreventable disaster.
-
For any function y = (x), the following hold true. (a) The real solutions of (x) = 0 correspond to the x-intercepts of the graph. (b) The real solutions of (x) < 0 are the x-values for which the...
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
(a) Write the propagation steps leading to the formation of dichloromethane (CH2Cl2) from chloromethane. (b) Explain why free-radical halogenations usually gives mixtures of products. (c) How could...
-
Draw resonance forms to show how the BHA radical is stabilized by delocalization of the radical electron over other atoms in the molecule.
-
The triphenylmethyl cation is so stable that some of its salts can be stored for months. Explain why this cation is so stable. triphenylmethyl cation
-
An investment offers the following cash flows: $554 today, $182 one year from now, $482 in 2 years, and $793 in 3 years. If the relevant interest rate is 7% per year (an APR, with interest compounded...
-
What is the internal rate of return (IRR) for a project that costs $5,500 and is expected to generate $1,800 per year for the next four years? If the firm's required rate of return is 8 percent, what...
-
Assume B Corporation has $375,000 of Ordinary Income from its operations, $75,000 of interest received from its investments on bonds from Z firm. Also, it received $50,000 of dividends from a...

Study smarter with the SolutionInn App


