Firefly luciferin is the compound that enables fireflies to glow. a. The structure exhibits three rings. Identify
Question:
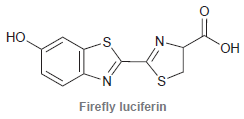
a. The structure exhibits three rings. Identify which of the rings are aromatic.
b. Identify which lone pairs are participating in resonance.
Transcribed Image Text:
Но ОН N. Firefly luciferin
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a b One of the lone ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Exposing Photographic Film the light-sensitive compound on most photographic films is silver bromide, Ag Br. A film is "exposed" when the light energy absorbed dissociates this molecule into its...
-
Experiments during a recent summer on a number of fireflies (small beetles, Lampyridae photinus) showed that the average interval between flashes of individual insects was 16.3 s at 21.0oC and 13.0 s...
-
Compound A with molecular formula C6H10 has two peaks in its spectrum, both of which are singlets (with ratio). Compound A reacts with an acidic aqueous solution containing mercuric sulfate to form...
-
Correction, overproduction, inventory and motion are all examples of: a. Waste b. 5 S target areas c. Noise d. Value-added activities
-
What mistakes are first-time exporters most likely to make? Outline the steps a small company should take to establish a successful export program.
-
Compare and contrast anti-miRNA oligonucleotides (AMOs), locked nucleic acids (LNAs), and antagomirs, which may eventually be used to treat certain forms of cancer.
-
Visit the website of the marketing research company GMI (http://www.gmi-mr.com/) and write a brief report on the methods they use to ensure effective data processing for their clients.
-
How would you characterize the decision-making styles of the two committees that considered the enrollment management problem? Would you characterize either of these processes as more effective or...
-
In what ways does the symbiotic relationship between mentors and mentees catalyze the transfer of tacit knowledge and foster the cultivation of adaptive expertise necessary for thriving amidst rapid...
-
Structco Construction Company entered into a long-term construction contract at a fixed contract price of $4,900,000 on September 1, 20x4. Work has proceeded since that time with the following...
-
Identify which of the following compounds are aromatic. a. b. c. d. e. O.
-
Identify each of the following compounds as aromatic, nonaromatic, or antiaromatic. Explain your choice in each case. a. b. c. d. e. f. g. h. :N-H
-
Figure P.5.104, which purports to show an erecting lens system, is taken from an old, out-of-print optics text. Whats wrong with it? P.5.104
-
Use the advertisement shown in Figure 11.12 as a basis for answering Problems 14-20. Assume the current interest rate is \(1.7 \%\). Figure 11. 12 What is the present value of the \(\$ 420,000\)...
-
Pale Company was established on January 1, 20X1. Along with other assets, it immediately purchased land for $80,000, a building for $240,000, and equipment for $90,000. On January 1, 20X5, Pale...
-
Continuing to use the annual reports of companies which you obtained for Chapters 1 and 4 , find the financial statements and the notes to the accounts. 1 Compare the financial statements with the...
-
In how many ways could a club of 15 appoint a committee of 4 people?
-
Salt Company, a newly established subsidiary of Pepper Corporation, received assets with an original cost of $260,000, a fair value of $200,000, and a book value of $140,000 from the parent in...
-
In problem, find the exact value. Do not use a calculator. sin 11/2
-
A police officer pulls you over and asks to search your vehicle because he suspects you have illegal drugs inside your car. Since he doesn't have reasonable suspicion to search your car, legally he...
-
Complete the following reactions. If no reaction is tikety, explain why. (a) (b) CH,SH + NaOH -_, (1 equiv.) 25 C CH OH
-
Outline a synthesis of each ether using either alcohol dehydration or alkene addition, as appropriate. (a) 2-methoxy-2-methylbutane (b) dibutyl ether
-
Give the structure of the that would with mCPBA to give each of the following expoxides. (a) (b) . /A C CH2 H,C C-4 CH,
-
What are the advantages and disadvantages of utilizing PostgreSQL's advanced indexing techniques such as GiST and GIN?
-
discuss the implications and trade-offs of utilizing PostgreSQL's advanced security features such as row-level security (RLS) and column-level encryption in compliance-sensitive applications...
-
How does PostgreSQL's support for advanced full-text search capabilities through extensions like pg_trgm and tsearch improve search functionality and performance in applications requiring...

Study smarter with the SolutionInn App


