For each of the following alkenes, assign the configuration of the double bond as either E or
Question:
a.
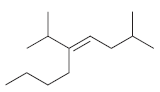
b.
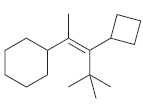
c.
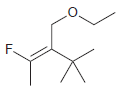
d.
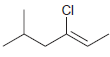
Transcribed Image Text:
F.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (17 reviews)
a...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The second-order rate constant (in units of M-1 s-1) for acid-catalyzed hydration at 25°C is given for each of the following alkenes: a. Calculate the relative rates of hydration of the alkenes....
-
Name each of the following alkenes or alkynes. a. CH2 = CH-CH2-CH3 b. c. d. e. f. g. CH3 C-CH-CH3 CH3 CH CH3 CH3CH2CH CH CH CH CH3 CH, C-CH-CH CH, CH2-CH, CH3 CH2CHs CH, CH2CH3 CH3 C C-CH CH3 CH3
-
Explain why each of the following alkenes is stable or unstable. (a) 1,2-dimethylcyclopentene (b) trans-1,2-dimethylcyclopentene (c) trans-3,4-dimethylcyclopentene (d) trans-1,2-dimethylcyclodecene...
-
In your opinion, was Saks' zero tolerance policy for employee theft reasonable? Was the policy likely cost-effective? Defend your answers.
-
What happens when a person has too much money? What about when an economy has too much money?
-
A rear window defroster consists of a long, flat wire bonded to the inside surface of the window. When current passes through the wire, it heats up and melts ice and snow on the window. For one...
-
The van der Waals equation of state is applicable for the (a) Solid phase only (b) Liquid phase only (c) Liquid and gas phases only (d) Solid, liquid and gas phases.
-
Deitz Manufacturing Inc. has three divisions which are operated as profit centers. Actual operating data for the divisions listed alphabetically are as follows. Instructions (a) Compute the missing...
-
a. Write a prototype for a function IsAlphaHigher that can be passed a pair of characters and that will return a Boolean value indicating whether or not the first character is higher alphabetically....
-
Draft the following sentences in active voice. The defendant was attacked by the plaintiff at the beginning of the argument. It is a requirement of good writing skills that active voice be used....
-
Which of the following best describes the efficiency of monopolistically competitive firms? a. Allocatively efficient by productively inefficient. b. Allocatively inefficient but productively...
-
Draw the mechanism and predict the product of the following reaction. In this case, H 3 O + must be used as a proton source instead of water. Explain why. 1) xs MeMgBr 2) H*
-
Asset Disposal Refer to Exercise 8-6. Assume that Gonzalez Company sold the asset on July 1, 2010, and received $15,000 cash and a note for an additional $15,000. Required 1. Identify and analyze the...
-
Inventory Turnover and days' sales in inventory Kracker Corp., Foodstuff, Inc., and Winston Stores, Inc. are three grocery chains in the United States. Inventory management is an important aspect of...
-
During the year ended 30 September 20x4 Hyper entered into two lease transactions: On 1 October 20x3 a payment of $90,000, being the first of five equal annual payments of a lease for an item of...
-
Write a C++ program to read any integer number and determine if the number is not prime using function int prime(), the answer will be printed from the main function.
-
What is the complexity of the following algorithm? input n for i 1 to 100 if n = i output "n = if n was not found output "n 100" output "done" "
-
The Dutch Water Authority wanted to raise money by selling perpetuities of $123 per year, with the first cash flow paid in one year from today. If the appropriate discount rate is 6.1%, what would...
-
Find the product. (z - 3) 3
-
Describe a group you belong or have belonged discuss the stages of group development and suggest how to improve the group effectiveness by using the group development model.
-
One of the two chair structures of cis-1-chloro-3-methylcyclohexane is more stable than the other by 15.5kJ/mol (3.7kcal/mol). Which is it? What is the energy cost of a 1, 3-diaxial interaction...
-
The German chemist J. Bredt proposed in 1935 that bicycloalkenes such as 1-norbornene, which have a double bond to the bridgehead carbon, are too strained to exist. Make a molecular model of...
-
Tell whether each of the following substituents on a steroid is axial or equatorial. (A substituent that is ?up? is on the top face of the molecule as drawn, and a substituent that is ?down? is on...
-
Part 1: The Program Explain in detail: The program you identified within your specialization area(My specialization is Special Education)that is in need of evaluation. Be sure to include the purpose...
-
Should the U.S. Government play a greater role in providing an economic "safety net" for its citizens than it currently does? Alternatively, should such a greater role be the obligation of...
-
How could I Identify and describe the segmentation of the workforce in a hospital setting (drawing up our study of how to differentiate employee groups depending on their drivers, such as flexible...

Study smarter with the SolutionInn App


