For each of the following compounds, determine whether the ring is activated or deactivated, then determine the
Question:
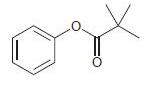
For each of the following compounds, determine whether the ring is activated or deactivated, then determine the strength of activation/deactivation, and finally, determine the expected directing effects.
(a)
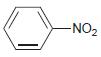
(b)
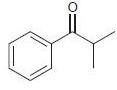
(c)
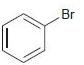
(d)
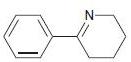
(e)
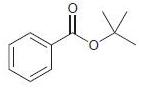
(f)
Transcribed Image Text:
-NO2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 41% (17 reviews)
a The nitro is strongly deactivating and metadirecting b An acyl group ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following compounds determine whether it exhibits a cis configuration or a trans configuration or whether it is simply not stereo-isomeric. a. b. c. d. e. f. g. F
-
For each of the following compounds, draw the important resonance forms. Indicate which structures are major and minor contributors or whether they have the same energy. (a) H2CNN (b) H2C=CH-NO2 (c)...
-
For each of the following compounds, give the systematic name and the common name (for those that have common names), and indicate whether the amines are primary, secondary, or tertiary: a. b. c....
-
For rigid-body motion, the strains will vanish. Under these conditions, integrate the straindisplacement relations (7.6.1) to show that the most general form of a rigid-body motion displacement field...
-
What is a unique selling proposition? What role should it play in a company's advertising strategy?
-
Sampson Corp. purchased a piece of equipment by issuing a \(\$ 20,000,6 \%\) installment note payable. Quarterly payments on the note are \(\$ 1,165\). What will be the reduction in the principal...
-
Bonds issued at a premium always have a. Interest expense equal to the interest payments b. Interest expense greater than the interest payments c. Interest expense less than the interest payments d....
-
Lynn Parsons is considering investing in either of two outstanding bonds. The bonds both have $1,000 par values and 11% coupon interest rates and pay annual interest. Bond A has exactly 5 years to...
-
Scott, S. E., Inbar, Y., & Rozin, P. (2016). Evidence for absolute moral opposition to genetically modified food in the United States. Perspectives on Psychological Science, 11(3), 315-324. This...
-
Data were collected on the amount spent by 64 customers for lunch at a major Houston restaurant. These data are contained in the file named Houston. Based on past studies the population standard...
-
Predict and explain the regiochemical outcome for chlorination of bromobenzene.
-
The following compound has two aromatic rings. Identify which ring is expected to be more reactive toward an electrophilic aromatic substitution reaction.
-
For each of the following misconceptions about the Fed, identify what is wrong with the statement and why. a. The Federal Reserve lacks accountability because no one audits the Fed. Theres no way to...
-
Explain the difference between increasing, constant and decreasing returns to scale.
-
Why is the point of profit maximization where marginal cost equals marginal revenue?
-
What are the characteristics of a competitive market? Which of the following drinks do you think is best described by these characteristics? Why arent the others? a. tap water b. bottled water c....
-
What do you think are the main costs of inflation that is out of control on the population?
-
Why can specialization and trade help improve standards of living?
-
The basic accounting equation is Assets = Liabilities + Owners equity. Replacing the words in that equation with dollar amounts, what is TSMCs accounting equation at December 31, 2016?
-
Solve the relation Exz:Solve therelation ne %3D
-
Two of the compounds given in Fig. Pl 1.78 form epoxides readily when treated with -OH, one forms an epoxid slowly, and one does not form an epoxide at all. Identify the compound(s) in each category...
-
Two of the compounds given in Fig. Pl 1.78 form epoxides readily when treated with -OH, one forms an epoxid slowly, and one does not form an epoxide at all. Identify the compound(s) in each category...
-
Two of the compounds given in Fig. Pl 1.78 form epoxides readily when treated with -OH, one forms an epoxid slowly, and one does not form an epoxide at all. Identify the compound(s) in each category...
-
In Year 1, Lee Incorporated billed its customers $56,300 for services performed. The company collected $41,700 of the amount bille Lee incurred $36,000 of other operating expenses on account. Lee...
-
The Mariana Trench is approximately 6.78 miles deep. Given that the specific gravity of sea water is 1.03, what is the hydrostatic (gauge) pressure at the bottom of the trench? What is the absolute...
-
All payments are end-of-period unless otherwise stated. a) The revenue stream of a project is estimated as $250K in the first year, and grows at 5% p.a. over the 15 years of the study period. MARR is...

Study smarter with the SolutionInn App


