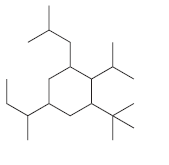
For each of the following compounds, identify all groups that would be considered substituents, and then indicate
Question:
(a)
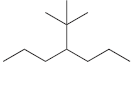
(b)
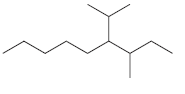
(c)
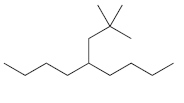
(d)
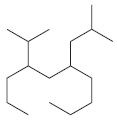
(e)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
a b c d e S...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Name each of the following compounds: (a) (b) (c) (d) (e) CH,CH NO2 di NO, CH2CH CI HO CH, OCH3 CH3 H3C CH(CH3)2 Cl 04 OCCCl3 Cl
-
Identify the common name for each of the following compounds: a. b. c. d.
-
Identify the functional groups in each of the following compounds: CH2CH2CH2CH2CH2CH3 Cl OH CH2CH2CH CH H2C CH3CH2CH2CH2 CH2CH2CH2CH3
-
Jillian operates a sporting goods store in a rented location at a shopping mall. She is insured under a CGL policy with the following limits: Indicate the dollar amount, if any, that Jillians insurer...
-
Watching an organization's failures play out on the news because they don't have a functioning crisis communications plan is both devastating and humiliating. Few things in corporate communications...
-
Ann is a self-employed restaurant critic who does her work exclusively from a home office. Anns income is $25,000 before the home office deduction this year. Her office takes up 200 square feet of...
-
P 55 Use the Standard Normal Table or technology to find the z-score that corresponds to the cumulative area or percentile. Table 4-Standard Normal Distribution Arca Z 0 Z .09 .08 .07 .06 .05 .04 .03...
-
In one of the little-known battles of the Civil War, General Tecumseh Beauregard lost the Third Battle of Bull Run because his preparations were not complete when the enemy attacked. If the critical...
-
The Hilbert matrix H is square matrix where row i and column j (using Python's zero indexing) of the n x n matrix are given by: 1 Aij for i = = 0,...,n 1, and for j = 0,...,n-1 i+j+1 For n=5 the...
-
Which of the graphs in Fig. Q25.12 best illustrates the current I in a real resistor as a function of the potential difference V across it? Explain. Figure Q25.12 (a) (b) (c) (d)
-
Derive the following expression for calculating the isothermal change in the constant volume heat capacity: (CV/V)T = T (2P/T2)V.
-
The following substituent is called a phenyl group: With this in mind, identify the systematic name for each substituent below: Phenyl
-
Define religion and promote religious inclusiveness. In addition, read the following definitions of religion and the scenario. You will use the scenario as the basis for this assessment. Three...
-
Information from a run ticket shows that 2,000 net barrels of oil with an API gravity of 36 were sold. The selling price is based on a contract price of $90/bbl, adjusted downward 4 for each degree...
-
Hays Company (a successful efforts company) owns 100% WI in a lease that is burdened with a 1/8 royalty interest owned by Alfred Smith. The undeveloped lease has capitalized costs of $40,000 and an...
-
Higgins Company, a full cost company located in Texas, sold 3,000 (gross) Mcf of gas with a heat content of 1.035 MMBtu/Mcf. The selling price of the gas was $9.00/MMBtu. REQUIRED: a. Determine the...
-
Bryant Oil Company produced a total of 1,000 barrels of oil in June 2022. The expected selling price was $120/bbl. The purchaser pays the severance taxes and the royalty interest owner and remits the...
-
A lease operated by Rocky Oil Company produces a total of 3,000 barrels of oil in June. The oil is sold in October. The posted field price and the actual selling price is $100/bbl. The severance tax...
-
How much is $1 million to be delivered 20 years in the future worth today if the interest rate is 20 percent?
-
Consider the reaction of acetic acid in water CH 3 CO 2 H(aq) + H 2 O(l) CH3CO 22 (aq) + H 3 O + (aq) where Ka 5 1.8 3 1025. a. Which two bases are competing for the proton? b. Which is the stronger...
-
Complete the molecular orbital description for the ground state of cyclopentadiene shown at right. Shade the appropriate lobes to indicate phase signs in each molecular orbital according to...
-
Rank the following dienes in order of increasing reactivity in a Diels-Alder reaction (1 = least reactive, 4 = most reactive). Briefly explain your ranking.
-
From each set of resonance structures that follow, designate the one that would contribute most to the hybrid and explain your choice: (a) (b) (c) (d) (e) (f) HO HO
-
A nursing team is having a routine meeting. One of the nurses, Stephen, is at the end of a 12-hour shift, and another nurse, Tanya, is just beginning hers. Tanya is a senior nurse in the unit with...
-
Assume that you are the leadership team of a convenience store chain that has more than 300 outlets. The company is facing an inventory shrinkage problem, and store managers report that the main...
-
1 . An employee at Amy's candles has noticed that some order quantities in the two reports presented are different although the order numbers are the same. We normally refer to such data as dirty....

Study smarter with the SolutionInn App


