For each of the following molecules, determine the number of carbon atoms present, and then determine the
Question:
a.
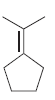
b.
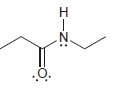
c.
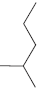
d.
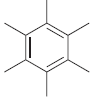
e.
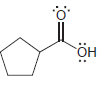
f.
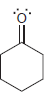
g.
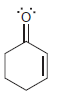
h.
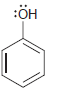
i.
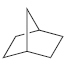
j.
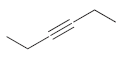
k.
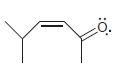
l.
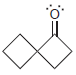
Transcribed Image Text:
-z:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
a b c d e f g ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following molecules that contain polar covalent bonds, indicate the positive and negative ends of the dipole, using the symbol v. Refer to Table 1.2 as needed. (a) HCI (c) HI (e) HOCI...
-
For each of the following molecules, state the bond angle (or bond angles, as appropriate) that you would expect to see on the central atom based on the simple VSEPR model. Would you expect the...
-
For each of the following molecules, state the bond angle (or bond angles, as appropriate) that you would expect to see on the central atom based on the simple VSEPR model. Would you expect the...
-
Debbie plans to buy a house for cash instead of paying a mortgage. She is willing to set aside $12 000 at the end of each year for 15 years. She puts her savings in a Tax Free Savings Account (TFSA)...
-
Define the velocity of money. What determines velocity? How are changes in velocity related to inflation/deflation?
-
A unity feedback system with the open loop transfer function \(\mathrm{G}(s)=\frac{1}{s(s+2)(s+4)}\) has gain margin of (a) \(33.6 \mathrm{~dB}\) (b) \(32.4 \mathrm{~dB}\) (c) \(32.6 \mathrm{~dB}\)...
-
Liberty's stock has traded recently around \(\$ 44\) per share. Use your answer to question 9 to measure the company's price/earnings ratio. a. 36 b. 44 c. 1.00 d. 69
-
Equivalence of various forms of Fick's law for binary mixtures (a) Starting with Eq. (A) of Table 17.8-2, derive Eqs. (B), (D), and (F). (b) Starting with Eq. (A) of Table 17.8-2, derive the folowing...
-
The marketing division of a large firm has found that it can model the sales generated by an advertising campaign as S(u) = 0.25u+ 1.5 millions of dollars when the firm invests u thousand dollars in...
-
Create two tables based on the E-R model shown in Figure 4-41 and the business rules in the following list for a work order tracking database. Include all the constraints in the CREATE TABLE...
-
Below are mass spectra for four different compounds. Identify whether each of these compounds contains a bromine atom, a chlorine atom, or neither. a. b. c. d. 100- 80- 60- 60- 40 20- 0- 60 70 10 20...
-
In Section 4.2, we learned how to name bicyclic compounds. Using those rules, together with the rules discussed in this section, provide a systematic name for the following bicyclic compound:
-
Prove that for any u and v in an inner product space V ||u||2 + ||u - v||2 = 2||u||2 + 2||v||2 Give a geometric interpretation of this result for the vector space R2
-
How has the world changed since two years ago (many possible answers here; which are most important)? How might these changes affect managers decisions?
-
What is a risk-adjusted discount rate (RADR) and when should a firm use one rather than its cost of capital to calculate an investments NPV?
-
Do you think gender affects leadership styles? If so, how? What would convince you to change one or more of your opinions?
-
Sticking with team sports, what examples can you think of in which high or low cohesiveness affected performance? And how much more or less do you think cohesiveness matters depending on the sport:...
-
It is 22 April 2015 and you are completing your taxes, albeit late, for 2014. You purchased your house for $600,000 on 1 January 2010 and financed 80% at 5.0% for 30 years using a constant payment...
-
Below is a payroll sheet for Otis Import plc for the month of September 2020. Assume a 10% income tax rate for all employees and an 8% Social Security tax on employee and employer. Instructions a....
-
A seasonal index may be less than one, equal to one, or greater than one. Explain what each of these values would mean.
-
Identify each pair of relationships among the ? OH groups in glucose (red?blue, red?green, red?black, blue?green, blue?black, green?black) as cis or trans. CH2OH OH OH Glucose OH
-
A Draw 1, 3, 5-trimethy1cyclohexane using a hexagon to represent the ring. How many cisTrans stereoisomers are possible?
-
From the data in Figure and Table, estimate the percentages of molecules that have their substituents in an axial orientation for the following compounds: (a) Isopropylcyclohexane (b)...
-
Magic Realm, Incorporated, has developed a new fantasy board game. The company sold 40,500 games last year at a selling price of $69 per game. Fixed expenses associated with the game total $729,000...
-
CALCULATE THE INITIAL COSTS, CALCULATE THE OPERATING CASH FLOW, CALCULATE THE TERMINAL NON OPERATING CASH FLOW, CALCULATE THE NPV . Initial Equipment $ 6 5 , 0 0 0 Project Life 3 Years Sales$ 5 5 ,...
-
A description of how each of the two management styles selected distinctively create organizational cultures. Describe the strengths and weaknesses of the two styles. Explain how these differences...

Study smarter with the SolutionInn App


