For each of the following pairs of compounds, identify the compound that would have the higher heat
Question:
(a)
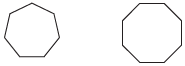
(b)
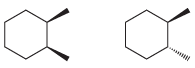
(c)

(d)

Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (18 reviews)
a has more CH 2 groups b cannot adopt a chair conformat...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following pairs of compounds, give one absorption band that could be used to distinguish between them: a. b. c. d. e. f. CH3CH2CH2CH and CH3CH2OCH3 CH CH2COCH and CH3CH2COH CH3CH2COH...
-
For each of the following pairs of compounds, identify one IR absorption band that could be used to distinguish between them: a. b. c. d. e. f. g. h. i. cis-2-butene and trans-2-butene j. CH3CH2CH2OH...
-
For each of the following pairs of compounds, indicate the compound that you would expect to be a more potent inhibitor of dihydrofolate reductase: a. b. NH NH CI of CH3 CH3 NH2 CH3 or H.NNCHCHCHCH,...
-
Use any method to determine if the series converges or diverges. Give reasons for your answer. n10 10" n=1
-
Explore the Internet to find a multinational company that has products that you enjoy building new consumer products for. Imagine that you are the marketing manager in charge of new products. The...
-
In an engine working on Otto cycle, air has a pressure of 1.0 bar and temperature \(30^{\circ} \mathrm{C}\) at the entry. Air is compressed with a compression ratio of 6 . The heat is added at...
-
Estimate the overnight radiant cooling possible from an open, \(30^{\circ} \mathrm{C}, 8 \mathrm{~m}\) diameter water tank during July in Chicago. What would you expect for convective and evaporative...
-
a. If Montreal wants to pursue the objective of minimizing the distance the snow must be moved (and therefore the cost of removing snow), how much snow should it plan to move from each sector to each...
-
Fastball (FB), a teenager who became a superhero after catching a radioactive baseball, can fly, and she can throw any object as fast as a jet aircraft with no air resistance at all. Right now FB is...
-
Slater & Gordon (S&G) became the worlds first listed law firm in 2007. The company is headquartered in Melbourne, where it was founded in 1935, by William Slater and Hugh Gordon....
-
Draw the ring flip for each of the following compounds: (a) (b) (c) OH I CI
-
For each of the following compounds determine whether it exhibits a cis configuration or a trans configuration or whether it is simply not stereo-isomeric. a. b. c. d. e. f. g. F
-
A company laid off one-sixth of its workforce because of falling sales. If the number of employees after the layoff is 690, how many employees were laid off?
-
a. Why is it important to have a good credit score? List at least 3 reasons. b. List the 5 factors that influence your credit score. c. What is a credit reporting bureau? (You may need to do some...
-
What PowerPoint slides should be included for HP Inc.'s audit program for the financing and investing process?
-
Who of the following were required to pay income tax under the first income tax legislation by Congress under the Sixteenth Amendment?
-
Consider the job you currently have in criminal justice or would like to have in the future. How might learning various budgetary approaches make you valuable to a criminal justice employer?
-
Once actual numbers from a physical inventory count are returned, what will the accounting department do with the information?
-
Graph the solution set of the system of inequalities. y 3 x y 2
-
Find the center of mass of a thin triangular plate bounded by the y-axis and the lines y = x and y = 2 - x if (x, y) = 6x + 3y + 3.
-
When benzaldehyde reacts with a peroxy acid, the product is benzoic acid. The mechanism for this reaction is analogous to the one just given for the oxidation of acetophenone, and the outcome...
-
Which compound in each of the following pairs has the higher boiling point? (Answer this problem without consulting tables.) (a) Pentanal or 1-pentanol (b) 2-Pentanone or 2-pentanol (c) Pentane or...
-
Give the structure of the product that would result from a Baeyer-Villiger oxidation of cyclopentanone.
-
Develop a 3-year financial projection for your proposed cancer tele-genetics program . State any and all assumptions you make in creating your estimates. Please consider the following in creating...
-
Lets assume that we have project L, which has a 40% chance of turning out good. The company Raises $7,000, 50% from bondholders demanding a 6% interest rate and 50% from stockholders, which becomes...
-
Please assist in the filling out the above and below table. I have provided all information that has been provided to me. Company Shares Outstanding Latest Share Price MVE Total Debt W d W e a b c =...

Study smarter with the SolutionInn App


