For each pair of compounds below, predict which compound will have the higher boiling point and explain
Question:
a) CH3CH2CH2OCH3 or CH3CH2CH2CH2OH
b) CH3CH2CH2CH3 or CH3CH2CH2CH2CH3
c)
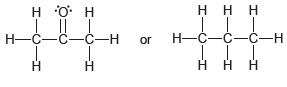
Transcribed Image Text:
ннн Н or С—Н Н—С—С—С—Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
a The second because it po...View the full answer

Answered By

Charles Okinda
students should give all the instructions concerning the challenge that they face. they will get an immediate response because I am always online.
4.90+
753+ Reviews
1481+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each pair of compounds below, determine whether they are identical compounds, constitutional isomers, stereoisomers, or different conformations of the same compound: (a) (b) (c) (d) (e) (f) (g)...
-
For each pair of compounds below, identify the more acidic compound: (a) (b) (c) (d) (e) (f) (g) (h) SH
-
For each pair of compounds below, identify the more acidic compound: (a) (b) (c) (d) (e) (f) (b) C (d) H. -S -3- (f) X H' `H.
-
In 2006, Walmart was operating more than 6,500 stores with 1.8 million employees around the world. In the early 1990s, Walmart Stores Inc. expanded into Canada, with the purchase of 122 stores from...
-
Based on, Kerzner, Harold. (2013) Project Management: A Systems Approach to Planning, Scheduling, and Controlling, 11th Ed. John Wiley & Sons. ISBN 978-1118022276.(Chapter 21) & Kerzner, Harold....
-
1. Which of the following taxpayers may not deduct their educational expense? a. A CPA who attends a course to review for the real estate agents exam b. A corporate president who attends a management...
-
.Find the indicated z-score(s) shown in the graph. Area = 0.5987 0 z = ? Z
-
At year-end 2013, the trial balance of Pennopscott Corp. showed the following accounts and amounts: Assume that, taken together, the variances are believed to be significant. Prepare the journal...
-
QUESTIONS: (1) This question is about the market for peanut butter and asks about the per-unit tax . (a) Sketch a graph of the market for peanut butter , labeling the supply and demand curves, both...
-
Determine VD for the fixed-bias configuration of Fig. 7.84. 18 V DSS 2 4 V FIG. 7.84 Problem 5.
-
Use the tabulated values of the enthalpy of combustion of benzene and the enthalpies of formation of CO 2 (g) and H 2 O(l) to determine H o f for benzene.
-
Derive the following expression for calculating the isothermal change in the constant volume heat capacity: (CV/V)T = T (2P/T2)V.
-
Describe how costs flow from inventory to cost of goods sold for the following methods: (a) FIFO and (b) LIFO.
-
Craig Petroleum has been operating in the United States for 15 years. The company purchased new tubing and casing to replace damaged tubular goods in a well that has been producing for four years....
-
Mountain Oil Company owns a 100% WI in a lease in Wyoming. The lease is burdened with a 1/5 royalty. During the month of July, a total of 10,000 barrels of oil was produced and sold. Assume the...
-
Mr. Stephens sold the surface rights and retained the mineral rights in some land in Texas. He leases the land to George Oil Company, reserving a 1/5 royalty. During 2019, George Oil Company makes...
-
Libby Oil Company has a working interest in a remote lease located in Wyoming. Libby agreed to pay the royalty owner a minimum royalty of $500/month in addition to the 1/5 royalty. Gas production on...
-
Garcia Company has a 100% WI in a lease in a remote area of Wyoming. The lease provides for a 1/5 royalty. Garcia produces and sells a total of 150,000 Mcf of gas from the property during August. Of...
-
Are dominant strategy equilibria always Nash equilibria? Are Nash equilibria always dominant strategy equilibria?
-
Using the information in P11-2B, compute the overhead controllable variance and the overhead volume variance. Data From Problem 11-2B: Huang Company uses a standard cost accounting system to account...
-
Dehydrohalogenation of 1,2-dihalides (with the elimination of two molar equivalents of HX) normally leads to an alkyne rather than to a conjugated diene. However, when 1,2-dibromocyclohexane is...
-
The heat of hydrogenation of allene is 298 kJ mol-1, whereas that of propyne is 290 kJ mol-1. (a) Which compound is more stable? (b) Treating allene with a strong base causes it to isomerize to...
-
Although both 1-bromobutane and 4-bromo-1-butene are primary halides, the latter undergoes elimination more rapidly. How can this behavior be explained?
-
Discuss the cultural differences between China and the United States. What would be the biggest adjustments I might need to make if working there as an expatriate? What are the most...
-
In her Ted talk, Kristi Rogers talks about the future of advertising and why it's crucial for ads to be relevant. She points out that even though we have lots of data and technology for digital ads,...
-
Consider ways in which an employer can make an employee feel like part of the team and/or empower them to act. More specifically, please respond to the following questions: How can an employer work...

Study smarter with the SolutionInn App


