For each pair of compounds, identify the stronger base. (a) (b) (c) vs. N. N. z. Vs.
Question:
(a)
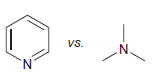
(b)
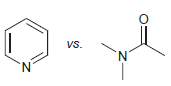
(c)
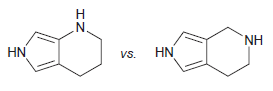
Transcribed Image Text:
vs. N. N. z. Vs. %3D N. N.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
a...View the full answer

Answered By

Rohith Bellamkonda
I am studying in IIT Indore,the most prestigious institute of India.I love solving maths and enjoy coding
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each pair of compounds, explain which is the stronger acid?
-
For each pair of compounds, describe which instrumental technique (IR, MS, proton NMR, carbon NMR) you could use to distinguish for certain which of the two compounds was in a sample. Describe what...
-
For each pair of compounds below, identify the more acidic compound: (a) (b) (c) (d) (e) (f) (g) (h) SH
-
If M is the midpoint of XY, find the coordinates of Y when X and M have the following coordinates: X(-4,2), M(0,3) Please write formulas too
-
1) Calculate the indicated amounts and financial ratios for Big Apple Enterprises. 3) ___43000__What is the company's Gross Profit in 2011? 4) __________What is the company's Gross Margin in 2011? 5)...
-
For the scaling numbers \[\begin{aligned}& \alpha_{1}=\frac{1+\sqrt{3}}{4 \sqrt{2}} \\& \alpha_{2}=\frac{3+\sqrt{3}}{4 \sqrt{2}} \\& \alpha_{3}=\frac{3-\sqrt{3}}{4 \sqrt{2}} \\&...
-
Have you experienced artistic portrayals of your own culture or identity that you find offensive? If so, do you think they should be censored? The statue of President Theodore Roosevelt flanked by an...
-
Major League Bat Company manufactures baseball bats. In addition to its goods in process inventories, the company maintains inventories of raw materials and finished goods. It uses raw materials as...
-
Presented below is information related to plant assets and intangible assets at year-end on December 31, 2025 for Wildhorse Co.: Buildings Goodwill Patents $1,132,800 355,200 460,800 Land 374,400...
-
Aluminum fins (k = 170 W/m-K) with triangular profiles shown in Figure P13-42 are used to remove heat from a surface with a temperature of 200 oC. The temperature of the surrounding air is 10 oC. The...
-
Suppose you are a marketer for a U.S. manufacturer of pet supplies. Two top executives have proposed expanding the company by opening retail stores and marketing pets on-sitepuppies, kittens,...
-
Draw the structure of each of the following compounds: (a) N-Ethyl-N-isopropylaniline (b) N,N-Dimethylcyclopropylamine (c) (2R,3S)-3-(N,N-Dimethylamino)-2-pentanamine (d) Benzylamine
-
Draw a Lorenz curve showing the distribution of income for the five people in the following table. Name Lena David Steve Jerome Lori Annual Earnings $70,000 60,000 50,000 40,000 30,000
-
4. Assume the BSM assumptions hold. A non-dividend-paying stock has a spot price of 10, an expected return of 5% per year and a return volatility of 20% per year. The continuously- compounded...
-
Thames Health System has a target capital structure of 30 percent debt and 70 percent equity. The system's CFO has obtained the following information: (1) the before-tax yield to maturity on the...
-
York Health has $800,000 of debt outstanding, and it pays an interest rate of 10 percent annually on its bank loan. York's total revenues are $3,200,000; its average tax rate is 40 percent, and its...
-
Assume perfect capital markets. Roche Technology's stock has a beta of 1.2 and has an expected return of 12.5%. It is a zero-debt firm. Suppose it issues new risk- free debt with a cost of debt of 5%...
-
Krugman Technology is currently a zero-debt firm with a stock price of $7.50 per share and 20 million shares. Although investors currently expect Krugman to remain a zero-debt firm, Krugman plans to...
-
Evaluate the following integrals. d V4 -
-
We all experience emotions, but some people disguise their true feelings better than others. Do you think this is a helpful or harmful thing to do? Under what conditions do you think it would be most...
-
Show both conrotatory processes for the thermal electtrocyclec conversion of (2E, 4E) - 2,4 -hexadiene into 3,4- dimethylcyclobutene (Eq. 27.8 ). Explain why the two processes are equally likely.
-
Show both conrotatory processes for the thermal electtrocyclec conversion of (2E, 4E) - 2,4 -hexadiene into 3,4- dimethylcyclobutene (Eq. 27.8 ). Explain why the two processes are equally likely.
-
After heating to 200 °C, the following compound is converted 95% yield into an isomer A that can be hydrogenated o cyclodecane. Give the structure of A, including its stereo chemistry.
-
Superman Enterprises has just completed an initial public offering. The firm sold 4,700,000 new shares at an offer price of $17.00 per share. The underwritering spread was $0.78 a share. The firm...
-
ABV is a non-dividend paying stock whose current price is $50. Its volatility is 12%. Over each of the next two 6-month periods the stock price is expected to go up by 9% or down by 8%. The risk-free...
-
Assume you have a portfolio consisting of a $400,000 investment in stock A and a $600,000 investment in stock B. Suppose that the daily volatilities of these two assets are 2% and 1.5%, respectively,...

Study smarter with the SolutionInn App


