For each pair of the following compounds, identify which compound would be expected to react more rapidly
Question:
For each pair of the following compounds, identify which compound would be expected to react more rapidly with a nucleophile:
(a)
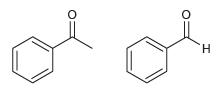
(b)
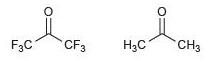
Transcribed Image Text:
H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
a...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each pair of the following compounds identify which compound would react more rapidly in an E1 reaction. a. b. CI .CI CI CI
-
For each pair of the following compounds identify which compound would react more rapidly in an E2 reaction: a. b. CI .CI Br Br
-
For each pair of the following compounds, identify which compound would react more rapidly in an S N 1 reaction. Explain your choice in each case. (a) (b) (c) (d) CI .CI
-
Holly funded the Holly Marx Trust in January 2020. The entire trust income is payable to her adult son Jack for 20 years. At the end of the twentieth year, the trust assets are to pass to Hollys...
-
On November 10, 2013, King Co. sold inventory to a customer in a foreign country. King agreed to accept 96,000 local currency units (LCU) in full payment for this inventory. Payment was to be made on...
-
Robert Brown first discovered the random motion of pollen immersed in water through a microscope. When seen with naked eyes, everything seemed to be still, but, upon viewing through a microscope, the...
-
Determine the displacement at point \(D\) of the overhang white spruce beam. A -4 ft. 150 lb/ft 300 lb D La -4 ft -B 3 in. H 16 in. Section a-a -4 ft- C
-
Diamond Jims makes and sells class rings for local schools. Operating information is as follows: Selling price per ring ........... $600 Variable cost per ring Rings and stones ........... $220 Sales...
-
What is at stake? Describe what you think is at stake with the issue of outsourcing ?
-
Assume you need to write and test a client-server application program on two hosts you have at home. a. What is the range of port numbers you would choose for the client program? b. What is the range...
-
Explain why the IUPAC name of a compound will never end with the suffix -1-one.
-
Draw the products of each Wittig reaction below. If two stereoisomers are possible, draw both stereoisomers: (a) (b) Ph Ph Ph-P Ph Ph. Ph-P Ph, Ph Ph
-
Suppose that Canada produces 1.0 million bicycles a year and imports another 0.4 million; there is no tariff or other import barrier. Bicycles sell for $400 each. * Parliament is considering a $40...
-
Discuss the role of the brain in controlling voluntary and involuntary bodily functions. How do different regions of the brain contribute to these functions?
-
In 2023, Jasmine and Thomas, a married couple, had taxable income of $154,000. If they were to file separate tax returns, Jasmine would have reported taxable income of $144,000 and Thomas would have...
-
In 2023, Sven is single and has $132,000 of regular taxable income. He itemizes his deductions as follows: real property tax of $2,600, state income tax of $4,600, and mortgage interest expense of...
-
Review the Microsoft Windows 10 Hardening GUidelines https://www.microsoft.com/security/blog/2019/04/11/introducing-the-security-configuration-framework-a-prioritized-guide-to-hardening-windows-10/...
-
Outline the profile of Microsoft windows Security management. Put together an outline profile of Microsoft windows Security management and explanation of how to be presented. Include any materials...
-
Use the Gauss-Jordan method to solve each system. 3x - 2y = 13 4x - y = 19
-
The financial statements of Eastern Platinum Limited (Eastplats) are presented in Appendix A at the end of this textbook. Instructions (a) Does East plats report any investments on its statement of...
-
Consider the cis and trans isomers of 1, 3-di-tert-butylcyclohexane (build molecular models). What unusual feature accounts for the fact that one of these isomers apparently exists in a twist boat...
-
Using the rules found in this chapter, give systematic names for the following or indicate that more rules need to be provided: (a) (b) (c) (d) Cl CI H C-B Br Br
-
Open the energy-minimized 3D Molecular Models on the book's website for trans-1-tert- utyl-3-methylcyclohexane and trans-1,3-di-tert-butylcyclohexane. What conformations of cyclohexane do the rings...
-
A description of how each of the two management styles selected distinctively create organizational cultures. Describe the strengths and weaknesses of the two styles. Explain how these differences...
-
A moon orbits a giant planet in a circular orbit with a semi-major axis a = 2.0473 x 10^8 metres and an orbital period P = 2.1299 days. What is the mass of the giant planet in kilograms to five...
-
Using the right-hand rule, in which direction will the magnetic force act on a positively charged particle that is moving to the left and experiencing a magnetic field straight ahead? a. Using the...

Study smarter with the SolutionInn App


