Hydroxide is not a suitable base for deprotonating acetylene: Explain why not. Can you propose a base
Question:
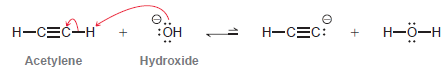
Explain why not. Can you propose a base that would be suitable?
Transcribed Image Text:
Н—СЕС-Н Н-СЕС: :Он Н-ӧ—н Acetylene Hydroxide
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
The equilibrium does not favor deprotonation of acetylene by hydroxi...View the full answer

Answered By

Madhur Jain
I have 6 years of rich teaching experience in subjects like Mathematics, Accounting, and Entrance Exams preparation. With my experience, I am able to quickly adapt to the student's level of understanding and make the best use of his time.
I focus on teaching concepts along with the applications and what separates me is the connection I create with my students. I am well qualified for working on complex problems and reaching out to the solutions in minimal time. I was also awarded 'The Best Tutor Award' for 2 consecutive years in my previous job.
Hoping to get to work on some really interesting problems here.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Can the function = x2 (L - 2x) be used as a trial wave function for the n = 1 state of a particle with mass m in a one-dimensional box of length L? If the answer is yes, then express the energy of...
-
Time-Dependent Wave Function for a Free Particle One example of a time-dependent wave function is that of a free particle [one for which U(x) = 0 for all x] of energy E and x-component of momentum p....
-
(a) Guanidine (shown) is about as strong a base as hydroxide ion. Explain why guanidine is a much stronger base than most other amines. (b) Show why p-nitroaniline is a much weaker base (3 pKb units...
-
Judy Baker is a new employee of Farnsdel and Babcock, LLP. Which of the following will provide proof of employment authorization for the completion of the I-9? 1. U.S. passport 2. U.S. military...
-
North Sea Oil has compiled the following data relative to current costs of its basic sources of external capitals, long-term debt, preferred stock, and common stock equity. Source of capital Cost...
-
This information relates to York Inc. for the year: Advertising expense ........... $ 2,400 Dividends paid ............. 7,000 Rent expense .............. 10,400 Retained earnings, January 1 ...........
-
Palanquin Auto is a national pre-owned car retailer that offers an online marketplace for customers interested in purchasing a preowned automobile. In an effort to reduce price negotiation, Palanquin...
-
On January 1, 2011, Argentina Corporation had the following stockholders equity accounts. Common Stock ($20 par value, 75,000 shares issued and outstanding) ................ $1,500,000 Paid-in...
-
Problem #4: Air at 100 kPa and 300 K flows steadily through a square duct with side length of 0.5 m. The air enters with a uniform speed of 5 m/s. An electric surface heating element is attached to...
-
Burger King is a US-based fast food restaurant chain. Burger King advertises, develops new foods, and procures ingredients, but it owns almost none of the actual restaurants. Instead, the restaurants...
-
Determine the position of equilibrium for each acid-base reaction below: (a) (b) (c) (d) `H. O + H20
-
Amino acids, such as glycine, are the key building blocks of proteins and will be discussed in greater detail in Chapter 25. At the pH of the stomach, glycine exists predominantly in aprotonated form...
-
Can you recall a time in which you used intuition to make a decision? Describe the situation and your intuitive moment.
-
Why do firms sponsor ADR or GDR programs?
-
Identify the new players in the global asset management industry.
-
What is the difference between sponsored and unsponsored American depositary receipts programs?
-
What are the key factors reshaping the asset management industry?
-
Compare the Sharpe ratio with the information ratio.
-
What does SQL stand for, and what purpose does it serve?
-
Economic feasibility is an important guideline in designing cost accounting systems. Do you agree? Explain.
-
How would the molecular ion peaks in the respective mass spectra of CH3Cl, CH2 Cl2, CHCl3, and CCl4 differ on the basis of the number of chlorines? (Remember that chlorine has isotopes 35Cl and 37Cl...
-
Provide the reagents necessary for the following synthetic transformations. More than one step may be required. (a) (b) (c) (d) (e) (f) OCH3 Br
-
Synthesize each of the following compounds by routes that involve allylic bromination by NBS. Use starting materials having four carbons or fewer. Begin by writing a retrosynthetic analysis. (a) (b)...
-
(i) When should a firm cut dividend pay out rate and increase retention rate to maximize share price? (ii) How can the dividend-discount model handle changing growth rates?
-
November 2021 (actual) December 2021 (budgeted) January 2022 (budgeted) Cash sales $ 80.000 Credit sales Total sales 240,000 $320,000 $100,000 360,000 $460,000 $ 60,000 180,000 $240,000 Management...
-
A firm has 200 shareholders, you among them. Each shareholder owns $20 worth of sto addition, Mr. Hostile owns 50 shares (for a firm total of 250 shares) and is trying to fi management. In an attempt...

Study smarter with the SolutionInn App


