Identify a systematic (IUPAC) name for each of the following compounds a. b. c. d. (e) CH
Question:
a.
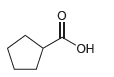
b.
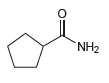
c.
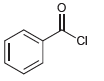
d.
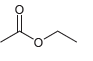
(e) CH3 (CH2)4CO2H
(f) CH3 (CH2)3COCl
(g) CH3 (CH2)4CONH2
Transcribed Image Text:
он O: NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
a Cyclopentanecarboxylic aci...View the full answer

Answered By

Sarah Khan
My core expertise are:
-_ Finance
-_ Business
-_ Management
-_ Marketing Management
-_ Financial Management
-_ Corporate Finance
-_ HRM etc...
I have 7+ years of experience as an online tutor. I have hands-on experience in handling:
-_ Academic Papers
-_ Research Paper
-_ Dissertation Paper
-_ Case study analysis
-_ Research Proposals
-_ Business Plan
-_ Complexed financial calculations in excel
-_ Home Work Assistance
-_ PPT
-_ Thesis Paper
-_ Capstone Papers
-_ Essay Writing etc...
5.00+
91+ Reviews
92+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write the IUPAC name for each of the following. a. b. c. d. CH CHCH2CHs H-C-OH CH3 ,.
-
Give the systematic name for each of the following compounds: a. b. c. d. e. f. g. h. NH
-
Give the systematic name for each of the following compounds: a. b. c. d. e. f. g. h. CH2CH3 CH3 CH2CH3 H3C CH3CHCH2CH2CH3 CH3CH2CHCH3 CH3CHCH3
-
In Problems 1130, solve each equation by factoring. x 2 + 4x = 0
-
Here is the topic "Creating long-term relationships" Business leaders are often troubled with unethical decisions. How do you plan to be a Christian business leader, while remaining loyal to your...
-
Western Digital Corporation (NYSE: WDC) manufactures hard disk drives. Exhibit 18 presents the companys consolidated balance sheet as of 29 March 2013. The balance sheet is labeled as unaudited...
-
Cumulative Poisson probabilities can be calculated using MINITAB. Output: Poisson with mean \(=1.64\) Find the cumulative Poisson probabilities for \(x=2\) and \(x=3\) when (a) \(\lambda=2.73\); (b)...
-
Where would each of the following items most likely be reported in a companys financial statements? Assume the monetary amount of each item is material and the company uses a periodic inventory...
-
Tiago makes three models of camera lens. Its product mix and contribution margin per unit follow: Lens A Lens B Lens C Required: Percentage of Unit Sales 30% 37 33 Contribution Margin per Unit $ 45...
-
Cost allocation to divisions. Forber Bakery makes baked goods for grocery stores, and has three divisions: Bread, Cake, and Doughnuts. Each division is run and evaluated separately, but the main...
-
Malonic acid has two acidic protons: The pKa of the first proton (pK 1 ) is measured to be 2.8, while the pK a of the second proton (pK 2 ) is measured to be 5.7. (a) Explain why the first proton is...
-
Identify the common name for each of the following compounds: a. b. c. d.
-
Pierre, a cash basis, unmarried taxpayer, had $1,400 of state income tax withheld during 2015. Also in 2015, Pierre paid $455 that was due when he filed his 2014 state income tax return and made...
-
Create a new class file with a main method. Using methods provided by the String class, try to print out the following output:
-
Customers of Dough Boy Ltd. have specified that pizza crusts they order should be 28-32 centimeters in diameter. Sample data recently collected indicate that Dough Boy's crusts average 30 centimeters...
-
(a) The three-hinged arch shown in Figure \(P 12.22\) has a parabolic profile. Draw the influence lines for both the horizontal and vertical reactions at \(A\) and the moment at \(D\). (b) Compute...
-
Suppose that a restaurant's requirements for the "4-ounce" hamburger patties that it purchased from its supplier should be between 3.4 and 4.6 ounces. Why would the restaurant be concerned about...
-
(a) Construct the influence lines for the moment and the vertical reaction \(R_{A}\) at support \(A\) for the beam in Figure P12.51. Evaluate the influence line ordinates at the quarter points of the...
-
How can a nucleus of uranium-235 be induced to fission? Describe what happens to the nucleus.
-
Three successive resonance frequencies in an organ pipe are 1310, 1834, and 2358 Hz. (a) Is the pipe closed at one end or open at both ends? (b) What is the fundamental frequency? (c) What is the...
-
What is the dissociation constant of an acid that has a pKa of (a) 4 (b) 7.8 (c) -2
-
Using the pka values in Table 3.1, calculate the equilibrium constant for the following reaction. F- acting as a base toward the acid HCN
-
For each of the following cases, give the product(s) of the transformation inducted by the curved - arrow notation. (a) (b) (CHs)2C-C(CH3)2 H-Br: O: O: -: 0 :
-
Design a power transmission system to connect a DC motor to its load. The motor is given in the motor catalogue as a BM1400 motor. You must create a transmission which will connect to this motor and...
-
SDJ, Incorporated, has net working capital of $3,320, current liabilities of $4,550, and inventory of $4,600. a. What is the current ratio? b. What is the quick ratio?
-
Calculate the final selling price to the nearest cent and markdown percent to the nearest hundredth percent: Original selling price $4,900 First markdown 20% Second markdown 10% Markup 12% Final...

Study smarter with the SolutionInn App


