Identify the Michael donor and Michael acceptor that could be used to prepare each of the following
Question:
(a)
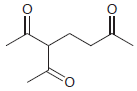
(b)
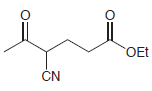
(c)
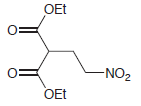
(d)
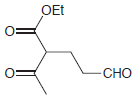
(e)
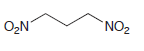
Transcribed Image Text:
OEt ČN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
a b c d e ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which diene and which dienophile could be used to prepare each of the following compounds? a. b. c. d. 1 CH3 Cl CI
-
Show how a conjugate addition can be used to prepare each of the following compounds. (a) 3,4-dimethyl-2-hexanone (2 ways) (b) levulinic acid
-
Show how a Wittig reaction can be used to prepare each of the following compounds. In each case, also show how the Wittig reagent would be prepared: (a) (b)
-
An orange grower in Florida faces a dilemma. The weather forecast is for cold weather, and there is a 50% chance that the temperature tonight will be cold enough to freeze and destroy his entire...
-
For the plane stress element shown in Figure P15 -14 subjected to a uniform temperature rise of T = 50°C, determine the thermal force matrix {fT}. Let E = 70 GPa, v = 0.3, α = 23...
-
Consider the following data for the airline industry for August 1, 2012 (EV = enterprise value, Book = tangible book value, NM = not meaningful because divisor is negative). Discuss the challenges of...
-
Eco-efficiency provides necessary but not sufficient indicators of environmental performance. What is environmental performance? Provide an example of another type of information that is important...
-
Discuss the factors used to classify retail establishments and list the types within each classification.
-
What role does the hypothalamic-pituitary-adrenal (HPA) axis and the sympathetic-adrenal-medullary (SAM) system play in the physiological stress response, and how can interventions target these...
-
Determine the resultant force acting on the 0.7-m-high and 0.7-m-wide triangular gate shown in Fig. P11??15 and its line of action. 0.3 m. 0.7 m 0.9 m Water 0.7 m
-
A(n) ______ ______ design is one in which each experimental unit is randomly assigned to a treatment. A(n) ______ ______ design is one in which the experimental units are paired up.
-
1. True or False: Observational studies are used to determine causality between explanatory and response variables. 2. True or False: Generally, the goal of an experiment is to determine the effect...
-
Prepare a cost of goods sold budget for Kenwood Manufacturing Inc. for the year ended December 31, 2013, from the following estimates. Inventories of production units: Direct materials purchased...
-
A telecommunications company provided its cable TV subscribers with free access to a new sports channel for a period of one month. It then chose a sample of 400 television viewers and asked them...
-
On what procedures does the auditor place heavy reliance in the audit of revenue and expense?
-
Distinguish between a reissued and an updated audit report.
-
Give two reasons for analyzing an expense account.
-
Describe an overall test of revenue for reasonableness.
-
What must be a company's dividend growth rate for its stock to have an expected value of $13.25, assuming its most recently paid dividend was $0.50 and the stock's required return is 10%?
-
Find an equation of the given line. Slope is -2; x-intercept is -2
-
Give the product formed when the following alcohols is oxidized by dilute HNO3. HOCH2CH 2CH2OH
-
Assuming you knew the properties of the compound obtained in Problem 24.23, including its optical rotation, show how you could use periodate oxidation to distinguish methyl -D- galactopyranoside from...
-
Assuming you knew the properties of the compound obtained in Problem 24.23, including its optical rotation, show how you could use periodate oxidation to distinguish methyl -D- galactopyranoside from...
-
Cash Supplies Land Building Total assets TYPHOON, INCORPORATED Balance Sheet December 31, 2024 Assets Liabilities $ 1,100 Accounts payable $ 3,400 Stockholders' Equity 5,400 Common stock 15,400...
-
DeSoto Tools Incorporated is planning to expand production. The expansion will cost $2,600,000, which can be financed either by bonds at an interest rate of 7 percent or by selling 52,000 shares of...
-
Waterway Inc. uses LIFO inventory costing. At January 1, 2025, inventory was $216,014 at both cost and market value. At December 31, 2025, the inventory was $283,252 at cost and $262,660 at market...

Study smarter with the SolutionInn App


