Identify the number of chirality centers in each of the following compounds: (a) (b) (c) `NH2 N'
Question:
(a)
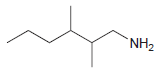
(b)
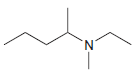
(c)
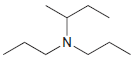
Transcribed Image Text:
`NH2 N'
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a T...View the full answer

Answered By

Moses mwangi
With prior writing experience, be sure that I will give a great grade, If not an A+, it will be something close to this. My reviews speaks it all, Try me!!
4.80+
78+ Reviews
157+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Name the following compounds: a. b. c. d. CH,CH OH Cl Br Br. CH,CH CH3
-
Predict the product(s) for each of the following reactions. In each case, make sure to consider the number of chirality centers being formed. a. b. c. d. e. f. Os0, (catalytic) NMO 1) OsO, 2) NaHSO,...
-
Name the following compounds: a. b. c. d. e. f. Br
-
Determine the degrees of freedom under the following conditions: (a) Tl-20 wt% Pb at 325 C and 400 C; (b) Tl-40 wt% Pb at 325 C and 400 C; (c) Tl-90 wt% Pb at 325 C and 400 C. Refer to the phase...
-
What is the internal rate of return for a project that has a net investment of $75,000 and the following net cash flows: Year 1 = $15,000; Year 2 = $20,000; Year 3 = $25,000; Year 4 = $30,000?
-
What are some of the advantages to using social media tools in the recruitment process?
-
Discuss the way teams develop over time.
-
The following facts pertain to a noncancelable lease agreement between Lennox Leasing Company and Gill Company, a lessee. Inception date: .................. May 1, 2012 Annual lease payment due at...
-
The Regal Cycle Company manufactures three types of bicycles-a dirt bike, a mountain bike, and a racing bike. Data on sales and expenses for the past quarter follow: Sales Variable manufacturing and...
-
Assume the same information as in P20.9. Instructions Follow the instructions assuming that McKee Electronics follows IFRS 16. Data From P20.9. The following facts pertain to a non-cancellable lease...
-
Do these findings surprise you? Why or why not? How might you apply what you have learned about social media and social media marketing from this chapter to your own workplace ethics?
-
1. Do you agree with P&G that online marketing will save money without hurting its brands in the market place? Why or why not? 2. What non-financial advantages of online marketing can P&G try to...
-
On January 1, 2022, Perriman Company transferred equipment for cash and leased it back. As seller-lessee, Perriman retained the right to substantially all of the remaining use of the equipment. The...
-
Kyle is the owner of a small business. He borrowed $1,000 from his local credit union and used 70% of it for his business and the other 30% for a family vacation. In 2023, he paid $100 in interest on...
-
Kirk decides to use the simplified option for his home office deduction on his 2023 tax return. He estimates that he uses 200 square feet of his home for the business. What is the allowable deduction...
-
In 2023, Tom is an insurance agent and small business advisor. He takes one of his clients to a college basketball game to guide him in a business transaction and sell him an insurance policy. Tom...
-
Megan is an independent lawyer. She performs legal services for her client, a small corporation. In payment for her services, Megan receives shares in the corporation with a market value of $5,000 at...
-
Nyah qualified as a bona fide resident of Spain for the years 2021, 2022 and 2023. She declares her income on a cash basis. In 2021 she was paid $70,000 for the work she did in Spain that year and in...
-
Describe a first step in integrating .3 X* x 2x + 4 dx.
-
What are some of the possible sources of information about a company that could be used for determining the companys competitive stance?
-
Ammonia appears in Table 1-5 both as an acid and as a conjugate base.
-
Write equations for the following acid-base reactions. Use the information in Table 1-5 to predict whether the equilibrium will favor the reactants or the products. (a) HCOOH + -CH (b) CH3COO- +...
-
Solved Problem 1-5(c) showed protonation of the double-bonded oxygen in acetic acid. Show the product of protonation on the other (-OH) oxygen. Explain why protonation of the double-bonded oxygen is...
-
MM Corp. has 50,000 shares outstanding with share price of $18. It has debt with market value of $300,000. The equity beta is 1.2 and debt beta is 0.1. The risk-free rate is 2% and the market risk...
-
The market is expected to return 15 percent next year and the risk-free rate is 7 percent. What is the expected rate of return on a stock with a beta of 1.3? The covariance of the market's returns...
-
A stock's current price is 145.05. A put option with an exercise price of 120 and maturity of 3 months is currently priced at $ 28.83. What is the option's time value?

Study smarter with the SolutionInn App


