Identify the reagents you would use to prepare the following compound via a Robinson annulation. . 0=
Question:
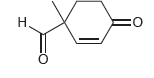
Transcribed Image Text:
Н. 0=
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
H g...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the reagents you would use to achieve the following transformation:
-
Identify the reagents you would use to accomplish the following transformation - Br Br
-
Identify the reagents you would use to achieve the following transformations: a. b. Br Br
-
At the beginning of the year, Plummer's Sports Center bought three used fitness machines from Advantage, Inc. The machines immediately were overhauled, installed, and started operating. The machines...
-
A company has the opportunity to do any, none, or all of the projects for which the net cash flows per year are shown below. The company has a cost of capital of 12%. Which should the company do and...
-
Layne Resources, Inc. has a $1,000 face value convertible bond outstanding that has a market value of $1,020. It has a coupon rate of 5 percent and matures in 12 years. The conversion price is $20....
-
A vapor bubble rises in a liquid. The relevant dimensional parameters are the liquid specific weight, \(\gamma_{\ell}\), the vapor specific weight, \(\gamma_{u}\), bubble velocity, \(V\), bubble...
-
GenX Furnishings Company manufactures designer furniture. GenX Furnishings uses a job order cost system. Balances on June 1 from the materials ledger are as follows:...
-
Jesse and Tim form a partnership by combining the assets of their separate businesses. Jesse contributes accounts receivable with a face amount of $45,000 and equipment with a cost of $179,000 and...
-
The Davenport Hotel, Inc., included the following stockholders' equity on its year-end balance sheet at December 31, 2016. Requirements 1. Identify the different issues of stock that the Davenport...
-
Draw a complete mechanism for the following transformation. NaOH, heat
-
Using cyclopentanone as your starting material and using any other reagents of your choice, propose an efficient synthesis for each of the following compounds. (a) (b) (c) OH HO.
-
The isotope: 32/15P is produced by the reaction; n +? 32/15P + p. what must be the target nucleus?
-
Blair orally promises Clay to sell him five crops of potatoes to be grown on Blackacre, a farm in Idaho, and Clay promises to pay a stated price for them on delivery. Is the contract enforceable?
-
Show that the power consumed by a 120-V device that draws 5 A of current is 500 W.
-
a. Ann owed \($2,500\) to Barry for services Barry rendered to Ann. The debt was due June 30, 2015. In March 2016, the debt was still unpaid. Barry was in urgent need of ready cash and told Ann that...
-
Refer to Exercise 7 and assume that the requirements are satisfied. Find the critical values that would be used to construct a 95% confidence interval estimate of . Exercise 7 Six human skulls from...
-
The zoology of emergence. In this chapter we listed the following examples of wellestablished classes of emergence: (1) Characteristic scale. (2) Collective degrees of freedom. (3) Transitions. (4)...
-
Two 20-penny nails are wrapped with insulated wire in the shape of a solenoid. They are then laid down end to end with the heads almost touching as shown in Figure Q20.16. If a battery is connected...
-
Explain how the graph of each function can be obtained from the graph of y = 1/x or y = 1/x 2 . Then graph f and give the (a) Domain (b) Range. Determine the largest open intervals of the domain over...
-
Treatment of acetic propionic anhydride with ethanol gives a mixture of two esters consisting of 36% of the higher boiling one, A, and 64% of the lower boiling one, B. Identify A and B.
-
Why are carboxylate salts much less reactive than esters in nucleophilic acyl substitution reactions?
-
You are employed by Fibers Unlimited, a company specializing in the manufacture of specialty polymers. The vice-president for research, Strong Fishlein, has asked you to design laboratory...
-
On a particular day, there were 300 stocks that advanced on the NYSE and 800 that declined. The volume in advancing issues was 1000 and the volume in declining issues was 3000. What is the trin ratio?
-
A coupon bond that pays interest of $30 annually has a par value of $1000, matures in 6 years, and is selling today at $850. What is the yield to maturity on this bond?
-
A callable bond pays annual interest of $40, has a par value of $1000, matures in 4 years but is callable in 3 years at a price of $1100, and has a value today of $1020. What is the yield to call on...

Study smarter with the SolutionInn App


