Identify which of the following methods is more efficient for producing 3,3 dimethylcyclohexene. Explain your choice. Br
Question:
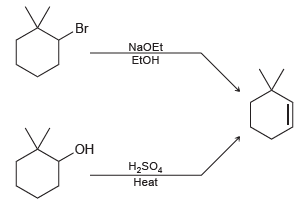
Transcribed Image Text:
Br NaOEt EEOH OH H,SO, Heat
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (13 reviews)
The first method is more efficient because it employs a strong ...View the full answer

Answered By

CHARLES AMBILA
I am an experienced tutor with more than 7 years of experience. I have helped thousands of students pursue their academic goals. My primary objective as a tutor is to ensure that students have easy time handling their academic tasks.
5.00+
109+ Reviews
323+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify which of the following monomers would be most reactive toward anionic polymerization: CH3 OAc CN
-
Identify which of the following monomers would be most reactive toward cationic polymerization. OAc CN CI
-
Identify which of the following functions are eigen functions of the operator d/dx: (a) D2/dx2, (b) Cos kx, (c) K, (d) Kx, (e) e-ax2. Give the corresponding eigen value where appropriate.
-
Discuss the interface between Purchasing and Supply Management and Logistics Management specifically with respect to selection of a third party logistics provider, analysis of the total cost of...
-
Prepare an analysis of the property rights, risks, and benefits of each in a minimum of, excluding the title and reference page, including the following: Decide what actions a manager in your...
-
Monique is a self-employed manufacturers representative (i.e., an independent contractor) who solicits business for numerous clients and receives a commission based on sales. She incurs the following...
-
How are Bode plots used?
-
Trayer Corporation has income from continuing operations of $290,000 for the year ended December 31, 2014. It also has the following items (before considering income taxes). 1. An extraordinary loss...
-
The contribution margin ratio of Kuck Corporation's only product is 62%. The company's monthly fixed expense is $455,000 and the company's monthly target profit is $41,000. Required: Determine the...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
Identify the pattern for each mechanism in Problem 8.34. For example the pattern for Problem 8.34a is: This mechanism is comprised of a proton transfer followed by the two core steps of an E1 process...
-
Calculate H and S if the temperature of 1.75 moles of Hg(l) is increased from 0.00 o C to 75.0 o C at 1 bar. Over this temperature range, C P,m = (J K -1 mol -1 ) 30.093 4.944 10 -3 T/K.
-
Draw the structure of histidine at pH 1, and show how the positive charge in the second basic group (the imidazole ring) can be delocalized.
-
Siphiwe sells handbags and shoes made from ostrich leather. Siphiwe's normal monthly sales are 200 handbags and 300 pairs of shoes. Handbags earn a contribution of R150, and each pair of shoes earns...
-
How do consolidated statements change "Subsequent to Acquisition"? Discuss the key considerations and adjustments that need to be made in the post-acquisition period. Elaborate on the methods and...
-
Brennan Corp. has provided the following account balances: Cash $38,100; Accounts receivable $6,100; Supplies $48,500; Long-term notes receivable $2,100; Equipment $96,500; Factory Building $181,000;...
-
The internal rate of return on a property purchased for $100,000 cash and generating $10.000 net annual cash flows for 10 years with a $ 100,000 net cash reversion expected at the end of the lease...
-
Given the following: 30-Day billing cycle and 1.5% interest charge per month. 9/1 Balance $1,500. 9/8 Charge $300. 9/20 Payment $1,000. What is the average daily balance on 10/01?
-
The factorial of a positive integer n can be computed as a product. n! = 1 2 3 g n Calculators and computers can evaluate factorials very quickly. Before the days of modern technology,...
-
Find the cross product a x b and verify that it is orthogonal to both a and b. a = (t, 1, 1/t), b = (t 2 , t 2 , 1)
-
What product(s) would you expect from the reaction of 1-methylcyclohexene with NBS? Would you use this reaction as part of asynthesis? CH NBS CCI4
-
How would you prepare the following compounds, starting with cyclopentane and any other reagents needed? (a) Chloro cyclopentane (b) Methylcyclopentane (c) 3-Bromocyclopentene (d) Cyclopentanol (e)...
-
Predict the product(s) of the followingreactions: SoCI. (b) CCH2H2CH2 () HBr Ether (d) (c) NBS PBr3 Ether Mg H20 A? (e) CH3CH2CHBrCH3 ? Ether Cul Li A? ? (f) CH3CH2CH2CH2BR Pentane Ether (g)...
-
A positive charge q = +8 nC is at the origin, and a second positive charge q = +12 nC is on the x axis at a = 4 m, Find the net electric field (a) at point P, on the x axis at x=7 m, and (b) at point...
-
8. Hooten Carpentry had the following accounts and account balances after adjusting entries. Assume all accounts have normal balances. Prepare the adjusted trial balance for Hooten Carpentry as of...
-
Explain at least one major difference that exists today between US GAAP and IFRS on the accounting for foreign currency transactions. Be specific.

Study smarter with the SolutionInn App


