In each case below, identify the Lewis acid and the Lewis base: (a) (b) CI CI .CI
Question:
(a)
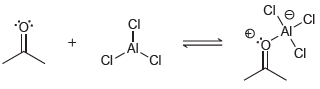
(b)
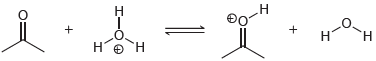
(c)
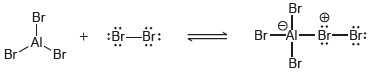
(d)

(e)
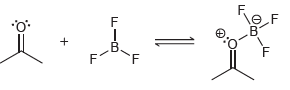
Transcribed Image Text:
CI CI .CI CI Н н н" TH. т Ф
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a b c d e O ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Identify the Brønsted-Lowry acid and base in the reaction (b) Identify the Lewis acid and base in the reaction. = H = N
-
Use the curved-arrow notation to write the reaction that would take place between dimethylamine (CH3)2NH and boron trifluoride. Identify the Lewis acid, Lewis base, nucleophile, and electrophile and...
-
In each reaction, identify the Lewis acid and the Lewis base: (a) (b) (c) F L-
-
27 13. Name-4 Abdominal Quadrants and 9 Abdominopelvic Regions using the PowerPoint Number Quadrant 1 2 3 4 Number Region 1 2 D. Bladder: E. Small Intestine: F. Spleen: G. Pancreas: 34 5 6 Tolla 7 8...
-
In a small manufacturing plant, the soldering iron shown below is used to solder connections on a large vertical panel. Several musculoskeletal injuries have been reported on this job over the last...
-
After a technical or managerial innovation, a firm can produce more today from a given amount of inputs than it could in the past. In farming, for example, new seed varieties, fertilizers, machines,...
-
If a reaction proceeds without loss or gain of heat and if all the products remain together in a single mass or stream of materials, these products will assume a definite temperature known as the (a)...
-
Ford 10-K a. What are the key revenue cycle accounts for Ford? What accounts involve critical accounting estimates? b. What does Ford say in Footnote 2 about its use of accounting estimates? What...
-
Graph the function. (3-x, x <0 h(x)= x +2, x0
-
The gas stream from a sulfur burner consists of 15 mol-% SO2, 20 mol-% O2, and 65 mol-% N2. The gas stream at atmospheric pressure and 400(C enters a catalytic converter A here 86% of the SO2 is...
-
List the following carbocations in order of increasing stability:
-
Provide an example of how treatment with cultured embryonic stem cells could be used to supply cells for the repair of damaged or diseased organs in human patients.
-
A machine acquired for 480,000 on 1 January 2006 and depreciated using the straight-line method, assuming 20 years life and 10% scrap value, was damaged in an accident on 1 January 2012. Though it...
-
What is the ending inventory using the average cost method? Fulbright Corporation uses the periodic inventory system. During its first year of operations, Fulbright made the following purchases...
-
Compute ABC Company's cost of capital based on the following information: Risk free rate: 2% Tax rate: 40% Effective interest of debt: 15% Market rate of return: 12% Beta: 1.6 FMV of preferred stock...
-
a. Which of the following is the first step in starting the research process? 1. ii. Searching sources of information to locate problem. Survey of related literature iii. Identification of problem...
-
A corporation has current assets of $3,203,000, current liabilities of $2,150,000, total assets of $10,000,000 and total liabilities of $6,000,000. If it pays $200,000 of its accounts payable what...
-
A particle (q= -4.0 C, m = 5.0 mg) moves in a uniform magnetic field with a velocity having a magnitude of 2.0 km/s and a direction that is 50 away from that of the magnetic field. The particle is...
-
Use the result of Exercise 84 to describe and graph the following lines. r(4 sin - 3 cos ) = 6
-
In muscle tissue, the ratio of phosphorylase a to phosphorylase b determines the rate of conversion of glycogen to glucose 1phosphate. Classify how each event affects the rate of glycogen breakdown...
-
Determine whether these structures represent the same compound or isomers; a) H-C. -C -C H-C-H H H b) H-C-C. H-C- H H H H-C-H H H TI C_CH C H H H c) H=C=C d) H-C-C H H-CH H-C- ...
-
Calculate the DU for these formulas and draw two constitutional isomer for each: (a) C 10 H 22 (b) C 9 H 16 (c) C 6 H 6
-
Convert these structures to skeletal structures: T a) H-C-C H H H-C-H C c) CH3CHCCHCH(CH3)2 C=C=C-H b) H H-C-H H H CH3 d) CH,CHCH,NHCH,CH CH,
-
Explain the concept of Remote Procedure Call (RPC) in IPC? How does it differ from traditional local IPC mechanisms, and what are its advantages in distributed systems?
-
Suppose we use the conventional grade point equivalent given in the following table. GPA Point Table Letter Grade point equivalent grade A 4 B 3 C 2 D 1 F 0 What is Amy's GPA at the end of the...
-
Consider the function y = log(x). A. What is the value of logs(3)?. B. What is the value of log(9)?, 8 C. What is the x-intercept of y = log(x)?. Why? 4 D. Use your answers to help sketch the graph...

Study smarter with the SolutionInn App


