In the compound below, identify all carbon atoms that are electron deficient (δ+) and all carbon atoms
Question:
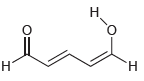
Transcribed Image Text:
Н н H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (18 reviews)
I qui 9...View the full answer

Answered By

Tobias sifuna
I am an individual who possesses a unique set of skills and qualities that make me well-suited for content and academic writing. I have a strong writing ability, allowing me to communicate ideas and arguments in a clear, concise, and effective manner. My writing is backed by extensive research skills, enabling me to gather information from credible sources to support my arguments. I also have critical thinking skills, which allow me to analyze information, draw informed conclusions, and present my arguments in a logical and convincing manner. Additionally, I have an eye for detail and the ability to carefully proofread my work, ensuring that it is free of errors and that all sources are properly cited. Time management skills are another key strength that allow me to meet deadlines and prioritize tasks effectively. Communication skills, including the ability to collaborate with others, including editors, peer reviewers, and subject matter experts, are also important qualities that I have. I am also adaptable, capable of writing on a variety of topics and adjusting my writing style and tone to meet the needs of different audiences and projects. Lastly, I am driven by a passion for writing, which continually drives me to improve my skills and produce high-quality work.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In each of the following compounds, identify all carbon atoms that you expect will be deficient in electron density (δ+). If you need help, refer to Section 1.5. a. b. c. Refer section...
-
The regions of + in a compound are the regions most likely to be attacked by an anion, such as hydroxide (HO - ). In the compound below, identify the two carbon atoms that are most likely to be...
-
Identify the number of carbon atoms and hydrogen atoms in the compound below:
-
The par yield curve for U.S. Treasury bonds is currently flat across all maturities at 5.50 percent. You have observed following "paired" transaction by your bond portfolio manager: Bond G H...
-
Design an algorithm that generates a maze that contains no path from start to finish but has the property that the removal of a prespecified wall creates a unique path.
-
The diameter of our Milky Way spiral galaxy is 100,000-180,000 light years and our solar system is approximately 25,000 light years from the galactic center. Recalling the effects of time dilation,...
-
Define the legal terms precedent, res judicata, stare decisis, original jurisdiction, and appellate jurisdiction.
-
The Snedecker Corporation is considering a change in its cash-only policy. The new terms would be net one period. Based on the following information, determine if the company should proceed or not....
-
The Assembly Department of the Long-Distance Golf Club Super Driver Company, has the following production and cost data at the end of March, 2021. Production: 21,800 units started into production;...
-
1. Does Brian have an obligation to lead the company to globalization? 2. What is Brians responsibility to himself and his family? 3. Consider Brians decision in light of rule-based, virtue, rights,...
-
The following reaction does not produce the desired product, but does produce a product that is a constitutional isomer of the desired product. Draw the product that is obtained, and propose a...
-
What role do domestic opportunity costs play in determining a nations area of comparative advantage and therefore specialization relative to that of a trading partner? Provide a numerical example (no...
-
With respect to the topics covered in this cha list and explain specifically two examples each w friction (a) Is desirable (b) Is not desirable.
-
Provide one (1) example of how being an inclusive and diverse may service impact the role of a service manager.
-
If an almond crop was sold on December 29, financial statements are prepared on December 31, and the check from the sale was received on January 5, when will the adjustment be recorded under the...
-
What percentage of adults is considered to be opinion leaders?
-
For the year ended, December 31, 2020, the following information is available for the three product lines that Kincaid, Inc. carries in its inventory: Product Cost Market R89 $108,500 $106,900 N83...
-
Errors in Inventory Count Bow Corp. accidentally overstated its previous year ending inventory by $950. Assume that ending current year inventory is accurately counted. The error in the previous year...
-
Nine voters are asked to rank three brands of potato chips: Pringles (P), Lays (L), and Kettle (K). The nine voters turn in the following ballots showing their preferences in order: Make a preference...
-
The ultimate goal of Google, Bing, and other consumer search engines is to provide users with search listings that contain useful information on the topic of their search. What recommendations would...
-
Why do you suppose no one has ever been able to make cyclopentyne as a stable molecule? Cyclopentyne
-
What is wrong with following sentences? The bonding molecular orbital in ethylene results from sideways overlap of two p atomic orbitals.
-
Allene, H2C = C = CH2, is somewhat unusual in that it has two adjacent double bonds. Draw a picture showing the orbitals involved in the and bonds of allene. Is the central atom sp2- or...
-
This project requires an investment in fixed assets, at time 0, of 44,000,000. Assets will be depreciated straight line in 5 years. This project has a 4 year life. The revenues for each of the 4...
-
Lorenzo Company applies overhead to jobs on the basis of direct materials cost. At year - end, the Work in Process Inventory account shows the following. Work in Process Inventory Date Explanation...
-
T-bill with a $10,000 par has 174 days until maturity. The T-bill has a bank discount bid quote of 2.208% and ask quote of 2.198%. What is the return on the investment in this T-bill if the trader...

Study smarter with the SolutionInn App


