In the next chapter, we will learn a method for preparing alkynes (compounds containing C ¡ C
Question:
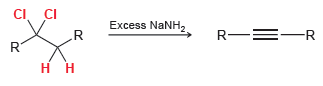
Transcribed Image Text:
CI. CI .R Excess NaNH, R-=-R R. R.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
CI CI...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
We?ll see in the next chapter that alkynes undergo many of the same reactions that alkenes do. What product might you expect from each of the following reactions? 1 equiv Br2 CH3 (a) 2 equiv H2, Pd/C...
-
Consider the structure of anisole (also called methoxybenzene). In the next chapter, we will discuss whether a methoxy group is electron donating or electron withdrawing. We will see that there is a...
-
A capacitor being charged has a current carrying charge to and away from the plates. In the next chapter we will define current to be dQ/dt, the rate of charge flow. What is the current to a 10 F...
-
Boeing Company was scheduled to deliver several of its 747-400 jumbo jetliners to Northwest Airlines by December 31, 1988. Northwest set that deadline because it needed the $16 million in investment...
-
Critical Thinking: A number of currency crises have affected certain countries, which have also resulted in contagion in the sense that the crises affected neighboring countries. In a critical essay,...
-
Explain how it is possible to have a deferred tax liability with regard to the presentation of a subsidiarys assets in a consolidated balance sheet, whereas on the subsidiarys balance sheet the same...
-
The following pressures for the air flow in Problem 4.24 were measured: Find the local rate of change of pressure \(\partial p / \partial t\) and the convective rate of change of pressure \(V...
-
Preparing a consolidated balance sheet. The first two columns of Exhibit 13.14 present information from the accounting records of Company P and Company S on December 31, 2009. Company P acquired l00%...
-
How does the selection of solvent polarity and solubility parameters influence the efficiency and selectivity of liquid-liquid extraction processes, particularly in the extraction of complex mixtures...
-
Which types of advisory services can be classified as a "pair of hands" role where you closely follow the clients instructions and are paid for effort?
-
Draw the mechanism for each of the following reactions: a. b. c. NaOMe CI NaOEt. Br
-
Identify whether each of the following reagents would be a strong nucleophile or a weak nucleophile, and also indicate whether it would be a strong base or a weak base: a.
-
(a) Find the inverse Laplace transform of (b) Solve using Laplace transforms the differential equation given that y = 3 when t = 0. S-4 s + 4s + 13
-
Write, compile, and simulate a VHDL description for the state machine diagram shown in Figure 6-38. Use a simulation input that passes through all paths in the state machine diagram, and include both...
-
We plan to remove about 90% of the A present in a gas stream by absorption in water which contains reactant B. Chemicals A and B react in the liquid as follows: B has a negligible vapor pressure,...
-
If the loop exits after executing only two iterations, draw a pipeline diagram for your MIPS code from 4.28.1 executed on a 2-issue processor shown in Figure 4.69. Assume the processor has perfect...
-
Using the references from Exercise 5.3, what is the miss rate for a fully associative cache with two-word blocks and a total size of 8 words, using LRU replacement? What is the miss rate using MRU...
-
At high pressure CO 2 is absorbed into a solution of NaOH in a packed column. The reaction is as follows: Find the rate of absorption, the controlling resistance, and what is happening in the liquid...
-
Find the derivative of the function in two ways: by using the Quotient Rule and by simplifying first. Show that your answers are equivalent. Which method do you prefer? 5x + Vx x* F(x) x?
-
What steps must a business take to implement a program of social responsibility?
-
Alkyl benzenes such as toluene (methylbenzene) react with NBS to give products in which bromine substitution has occurred at the position next to the aromatic ring (the benzylic position). Explain,...
-
Draw resonance structures for the benzyl radical, C6H5CH12, the intermediate produced in the NBS bromination reaction of toluene.
-
What product would you expect from the reaction of l-phenyl-2-hutene with NBS?Explain. 1-Phenyl-2-butene
-
Consider the seventh excited level of the hydrogen atom. A)What is the energy of this level? B)What is the largest magnitude of the orbital angular momentum? C)What is the largest angle between the...
-
1. How many degrees Celsius does the temperature decrease when there is a 45.8F decrease in temperature? 2. For each 1.00F change in temperature what is the corresponding change in degrees Celsius?...
-
A proton (mass of 1.67 x 10-27 kg) is moving at 1.25 x 106 m/s directly toward a stationary helium nucleus (mass 6.64 x 10-27 kg). After a head-on elastic collision, what is the velocity of the...

Study smarter with the SolutionInn App


