Lactones can be prepared from diethyl malonate and epoxides. Diethyl malonate is treated with a base, followed
Question:
![1) NaOEt EtO EtO OEt 2) OEt Н,о Heat [H,O*] Н,о* Heat Но Но ОН (-CO2) ОН ОН](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/images/question_images/1525/0/6/8/3845ae6b260351221525068355373.jpg)
Using this process, identify what reagents you would need to prepare the following compound:
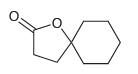
Transcribed Image Text:
1) NaOEt EtO EtO OEt 2) OEt Н,о Heat [H,O*] Н,о* Heat Но Но ОН (-CO2) ОН ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Eto OE...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Outline syntheses of each of the following from diethyl malonate and any other required reagents: (a) 2-Methylbutanoic acid (b) 4-Methyl-1-pentanol (c) (d) OH
-
Thiols can be prepared from the reaction of thiourea with an alkyl halide, followed by hydroxide-ion-promoted hydrolysis. a. Propose a mechanism for the reaction. b. What thiol would be formed if the...
-
When ethylene oxide is treated with a strong nucleophile, the epoxide ring is opened to form an alkoxide ion that can function as a nucleophile to attack another molecule of ethylene oxide. This...
-
aj b) If the magnitude of the force F is 700 N, what is the value of the internal moment at point A in N.m? d) 0.5 m 540 180 210 A 0.8 m 240 Bo birak F A
-
For the composite wall idealized by the one-dimensional model shown in Figure P13-7, determine the interface temperatures. For element 1, let Kxx = 5 W (m ( oC) for element 2, Kxx = 10 W (m ( oC);...
-
An intercom message is received stating that a hostile intruder situation exists in the building and that a shelterin- the place should be performed. Subsequently, a fire alarm is heard indicating...
-
A machine is subjected to the vibration \[x(t)=20 \sin 50 t+5 \sin 150 t \mathrm{~mm} \quad(t \text { in } \mathrm{s})\] An accelerometer having a damped natural frequency of \(80 \mathrm{rad} /...
-
Kathy Wintz formed a lawn service business as a summer job. To start the business on May 1, she deposited $1,000 in a new bank account in the name of the business. The $1,000 consisted of a $600 loan...
-
Bath Manufacturing Company uses the weighted average method for process costing. Bath produces processed food products that pass through three sequential departments. The costs for Department 1 for...
-
Sato Jewellers has had a request for a special order for 10 gold bangles for the members of a wedding party. The normal selling price of a gold bangle is $389.95 and its unit product cost is $264.00,...
-
Draw the structure of the product with molecular formula C 10 H 10 O that is obtained when the compound below is heated with aqueous acid. CN CN C10H100 Heat
-
Predict the major product of the following transformation. CO2ET C10H100 Heat
-
How does the Federal unified transfer tax differ from an income tax?
-
On July 1, 2023, Pharoah Corp. had outstanding 6%, $1006000, 10-year bonds maturing on June 30, 2030. Interest is payable semi-annually on June 30 and December 31. Assume all appropriate entries had...
-
Assume a corporation has 15,000,000 shares of common stock authorized, of which (i) 8,000,000 shares have been issued, (ii) 1,000,000 shares have been reserved for the conversion of convertible...
-
Explain what is meant by information asymmetry and provide and example.
-
W hat kind of risks are there in future, forward , option and swap contract? please answer each contract with their risks separately.
-
Malinin Company distributes a single product. The company's sales and expenses for the current month follow: Per Sales Variable expenses Fixed expenses Total $ 612,000 Unit $ 40 428,400 28...
-
A power line is attached at the same height to two utility poles that are separated by a distance of 100 ft; the power line follows the curve f(x) = a cosh(x/a). Use the following steps to find the...
-
For each equation, (a) Write it in slope-intercept form (b) Give the slope of the line (c) Give the y-intercept (d) Graph the line. 7x - 3y = 3
-
Outline a synthesis for each of the following compounds from the indicated starting material and any other reagents. 4-methyl-3-nitropyridine from - lpicoline
-
Predict the predominant product in each of the following reactions. Explain your answer. 3,4-dibromopyridine + NH3, heat (C5H5BrN2)
-
Using bases (B:) and acids (+BH) as needed, provide a curved-arrow mechanism for the conversion of the c-amino acid serine into formaldehyde and glycine (Eq. 25.53, p. 1242). Eq. 25.53 formaldehyde...
-
On Monday, April 5, 2010, just before 3:00 in the afternoon, miners at Massey Energy Corporation's Upper Big Branch coal mine in southern West Virginia were in the process of a routine shift change....
-
Ethics in Human Resource Practices HR teams will play vital roles in organizations to ensure ethical behavior and compliance. To be effective, they must first place an emphasis on ethical behavior...
-
Why do managers struggle with ethics when it comes to making strategic management decisions for their organization?

Study smarter with the SolutionInn App


