Match each compound with the appropriate spectrum. a. b. c. d. .CI OH Br ZHN
Question:
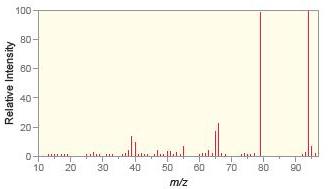
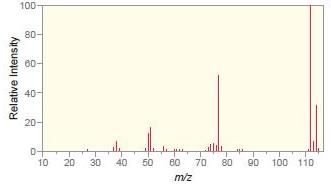
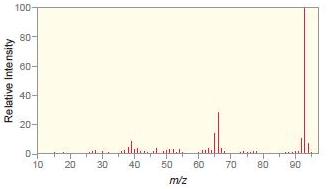
Match each compound with the appropriate spectrum.
a.
b.
c.
d.
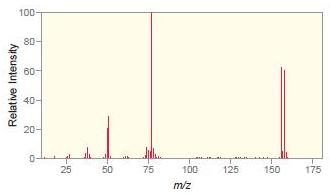
Transcribed Image Text:
.CI OH Br ZHN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
a ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Match each compound with the appropriate IR spectrum: a. b. c. d. e. f. `NH2 100- 60- 20- 4000 3500 3000 2500 2000 1500 1000 Wavenumber (cm-1) % Transmittance
-
Match the compound with the appropriate carbonyl IR absorption band: acyl chloride ........................ ~1800 and 1750 cm-1 acid anhydride ..................... ~1640 cm-1 ester...
-
Figures 12.15, 12.16, 12.17, and 12.18 are the IR spectra of four compounds: hexanoic acid, 1-pentanol, cyclohexane, and 3-pentanone, respectively. Match each compound with the correct spectrum,...
-
In Exercises 8194, begin by graphing the absolute value function, f(x) = |x| . Then use transformations of this graph to graph the given function. h(x) = x +31-2
-
What skills do project managers need now that they did not need in the past? What skills do you think project managers will need in the next 50 years that they do not have now? Provide a rationale...
-
A banking executive is studying the role of trust in creating customer advocates, and how valuable trust is to the overall banking relationship. Based on study results, the executive has determined...
-
Longitudinal vibration of an isotropic, particle-reinforced composite bar may be modeled by using the 1D wave equation (Equation 8.99) if the material is linear elastic. Derive the equation of motion...
-
John Ingles has provided you with the following information related to his various investment holdings as of December 31, 2012. Interest earned on joint bank account with his spouse (spouse...
-
CEOs often engage in earnings manipulation by increasing or decreasing their companies' earnings to meet their goals. Suggest any preventive measures to reduce CEOs' engagement in earnings...
-
Next let's estimate the capital cost of the solar system in dollars per kilowatt. Use Figure 7 from the US National Renewable Energy Laboratorys 2015 US Photovoltaic Prices and Costs Breakdown...
-
The mass spectrum of an unknown hydrocarbon exhibits an (M+1) peak that is 10% as tall as the molecular ion peak. Identify the number of carbon atoms in the unknown compound.
-
The sex attractant of the codling moth gives an IR spectrum with a broad signal between 3200 and 3600 cm -1 and two signals between 1600 and 1700 cm -1 . In the mass spectrum of this compound, the...
-
Using the method of intersecting each machine with the complement of the other, show that do not accept the same language. (1 (1 8. and (1 (1 h (1 + "
-
Why is granting credit riskier in an international context?
-
What are some factors that make cash management more complicated in a multinational corporation than in a purely domestic corporation?
-
Are there international differences in firms financial leverage? Explain.
-
Why do Eurobonds appeal to investors?
-
Differentiate between foreign bonds and Eurobonds.
-
Repeat Exercise 53 for a polynomial function with dominating term -9x 6 . Exercise 53 If the dominating term of a polynomial function is 10x 7 , what can we conclude about each of the following...
-
Burberrys competitive advantage is through its differentiation strategy. What risk should Burberry remain aware of?
-
(a)Construct a hybrid orbital picture for the hydronium ion (H3O+) using oxygen sp3 hybrid orbitals. (b) How would you expect the H-O-H bond angles in hydronium ion to compare with those in water...
-
(a)Construct a hybrid orbital picture for the hydronium ion (H3O+) using oxygen sp3 hybrid orbitals. (b) How would you expect the H-O-H bond angles in hydronium ion to compare with those in water...
-
Which of the atoms in each of the following species has a complete octet? What is the formal charge on each? Assume all unshared valence electrons are shown. (a) CH (b) :NH3 (c) :CH3 (d) BH3 (e) :T:...
-
Consider the vectors u=2ij2k and v=2i+2j+4k. Find the vector components of u that are parallel and perpendicular to v.
-
1. Determine whether the following sets form subspaces and justify your answers. (a) {(1, 2): 122 = 0} in R (b) {(1, 2) |1|-|x2|} in R2 (c) {(x1, x2, x3): x3 = 21 or x3 = 12} in R3 (d) {(x1, x2, x3):...
-
Solve the system of two equations for y two equations are: A1=ax+by A2=cx+dy

Study smarter with the SolutionInn App


