Name each of the following compounds: (a) (b) (c) (d) (e) (f) (g) (h) (i)
Question:
Name each of the following compounds:
(a)
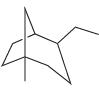
(b)
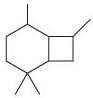
(c)
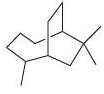
(d)
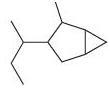
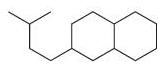
(e)
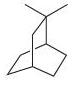
(f)
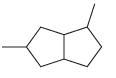
(g)
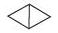
(h)
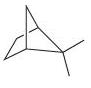
(i)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (18 reviews)
a 4ethyl1methylbicyclo 321 octane b 2257tetramethylbicycl...View the full answer

Answered By

Hassan Ali
I am an electrical engineer with Master in Management (Engineering). I have been teaching for more than 10years and still helping a a lot of students online and in person. In addition to that, I not only have theoretical experience but also have practical experience by working on different managerial positions in different companies. Now I am running my own company successfully which I launched in 2019. I can provide complete guidance in the following fields. System engineering management, research and lab reports, power transmission, utilisation and distribution, generators and motors, organizational behaviour, essay writing, general management, digital system design, control system, business and leadership.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Name each of the following compounds according to substitutive IUPAC nomenclature: (a) (CH3)2CHCH2CH2CH2Br (b) (CH3)2CHCH2CH2CH2OH (c) Cl3CCH2Br (d) (e) CF3CH2OH (f) (g) (h) (i) Cl2CHCHBr CI OH ,
-
Name each of the following compounds. a. CuI b. CuI2 c. CoI2 d. Na2CO3 e. NaHCO3 f. S4N4 g. SeBr4 h. NaOCl i. BaCrO4 j. NH4NO3
-
Name each of the following compounds: a. HOCH2CH(OH)CH(OH)CH2OH b. (CH3)3CO-K+ c. d. e. f. g. CH3CH = CHCH2OH h. i. CH3CH2CH(OH)CH3 j. CH3CHBrC(CH3)2OH OH Br ,
-
Find the inverse of each of the given matrices by the method of Example 1 of this section. Data from Example 1 Find the inverse of the matrix First, we interchange the elements on the principal...
-
Write a paper on any Ethnic group
-
Your supervisor has asked you to research the following situation concerning Aurora Browning. Aurora is a self-employed attorney practicing in Seattle, Washington. Aurora attended a legal convention...
-
P 95 Use the Standard Normal Table or technology to find the z-score that corresponds to the cumulative area or percentile. Table 4-Standard Normal Distribution Arca Z 0 Z .09 .08 .07 .06 .05 .04 .03...
-
1. What personality and behavior characteristics does Henry Rankin exhibit? Do you think these traits contribute to a good personjob fit for him? If you were an executive coach hired to help Rankin...
-
1. Create database named 'Watch'. Create a table named 'Watch' with the data fields as stated below in the database. Watch ID (Primary Key) Model Kind of watch glass Water resistance Strap Validation...
-
Priti is a trader. Her financial year ends on 31 March. Her trial balance on 31 March 204 was as follows: Additional information: 1. At 31 March 204: Inventory was valued at $41,050. Commission...
-
Draw a bond-line drawing for each of the following compounds: (a) 3-Isopropyl-2,4-dimethylpentane (b) 4-Ethyl-2-methylhexane (c) 1,1,2,2-Tetramethylcyclopropane
-
Draw a bond-line structure for each of the following compounds: (a) 2,2,3,3-Tetramethylbicyclo [2.2.1] heptane (b) 8,8-Diethylbicyclo [3.2.1] octane (c) 3-Isopropylbicyclo [3.2.0] heptane
-
The system acetone(1) + methanol(2) is well represented by the one-parameter Margules equation using A = 0.605 at 50C. (a) What is the bubble pressure for an equimolar mixture at 30C? (b) What is the...
-
Tequila Crisis (C) (web exercise). Log in to IMF International Financial Statistics to present and analyze Mexicos capital account over the period 1992 1996. How can you reconcile your findings with...
-
Bristols Bicycles maintains inventory records under the perpetual inventory system. At 30 June 2019, the inventory balance determined by the system showed a value of $300 000. However, on conducting...
-
Given the assumptions of a perfectly competitive industry, explain why firms operating in that industry are reluctant to invest in new technological development.
-
State the amount of revenue that should be recognised by Whitehall Ltd in the year ended 31 December 2020 for each item below, justifying your answer by reference to the revenue definition and...
-
Part A Branxton Ltds inventory on 1 April 2019 had a cost of $100 000 and a retail value of $170 000. During April, the companys net purchases cost $216 000 and had a net retail value of $324 000....
-
What is the present value of $100 one year from now if the interest rate is 10%? What is the present value if the interest rate is 5%?
-
Is it ethical to provide safety training in English to immigrant workers who speak little English, in order to reduce costs?
-
Mixing furan (Problem 13.45) with maleic anhydride in diethyl ether yields a crystalline solid with a melting point of 125(C. When melting of this compound takes place, however, one can notice that...
-
Draw the structure of the product from the following reaction (formed during a synthesis of one of the endiandric acids by K. C. Nicolaou): MeO2C osi(t-Bu)Phe toluene, 110C
-
Draw all of the contributing resonance structures and the resonance hybrid for the carbocation that would result from ionization of bromine from 5-bromo-1,3-pentadiene. Open the computer molecular...
-
Give a summary of concepts and the applications for the following quistion: When a 200.0-g mass is attached to a spring, it stretches the spring by 7.50 cm. With that mass-spring system in...
-
A helium-filled balloon, whose envelope has a mass of 0.29 kg, is tied to a 3.8-m long, 0.054-kg string. The balloon is spherical with a radius of 0.40 m. When released, it lifts a length h of the...
-
A potato is launched out of a building (building A) from the 10th floor which is 15 m high, with a velocity of 55 m/s at an angle of 35.0 above the horizontal. There is a building (building 2) 130 m...

Study smarter with the SolutionInn App


