Phosgene is highly toxic and was used as a chemical weapon in World War I. It is
Question:
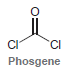
(a) When vapors of phosgene are inhaled, the compound rapidly reacts with any nucleophilic sites present (OH groups, NH2 groups, etc.), producing HCl gas. Draw a mechanism for this process.
(b) When phosgene is treated with ethylene glycol (HOCH2CH2OH), a compound with molecular formula C3H4O3 is obtained. Draw the structure of this product.
(c) Predict the product that is expected when phosgene is treated with excess phenylmagnesium bromide, followed by water.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





