Predict the major product for each of the following transformations: (a) 1) Br2, PB13 2) H20
Question:
(a)
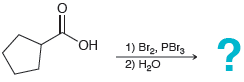
(b)
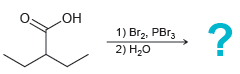
Transcribed Image Text:
"ОН 1) Br2, PB13 2) H20 но 1) Br2, PBR3 2) H20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
a b OH ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product for each of the following transformations. (a) (b)
-
Predict the major product for each of the following reactions: (a) (b) [H') NH3 -H20 NH2 [H*)-H20
-
Predict the major product for each of the following reactions: (a) (b) [H*] N-H -H20 :? [] -H20
-
If you deposit $2,000 today into an account earning an annual rate of return of 9 percent, what would your account be worth In 30 years? a. If you deposit $2, 000 today into an account earning an...
-
XZYY, Inc. currently has an issue of bonds outstanding that will mature in 24 years. The bonds have a face value of $1,000 and a stated annual coupon rate of 17% with annual coupon payments. The bond...
-
Do you think that long-run economic growth is simply a matter of having higher wages, and at the same time, having more of the same goods and services, or would you say that it entails other...
-
Wendy Craven is the sole shareholder of a property management company near the campus of Pensacola Junior College. The business has cash of \($6,000\) and furniture that cost \($12,000\) and has a...
-
Closing Journal Entries. At the end of a fiscal year, budgetary and operating statement control accounts in the general ledger of the General Fund of Dade City had the following balances:...
-
Here is the homework example we used in CH 10 for current rate method. I have made a change on the exchange rate on 1/1/2010 (from 0.50 to 0.52). Everything else remains the same. Please make...
-
The trim line at PW is a small subassembly line that, along with other such lines, feeds into the final chassis line. The entire assembly line, which consists of more than 900 workstations, is to...
-
Identify the reagents that you would use to accomplish each of the following transformations: (a) (b) H.
-
Go to www.victoriassecret.com. How does this famous website use enhancement of the self to attract consumers? Would you expect males to visit the site as well as females? How could the site make it...
-
A tax of $8 per unit is imposed on the supplier of an item. The original supply curve is q = 0.5p 25 and the demand curve is q = 165 0.5p, where p is price in dollars. Find the equilibrium price...
-
Differentiate between an automatic controller and process controller.
-
What is System I&CO?
-
What is operational site activation and what is its scope?
-
What is a sensitivity analysis?
-
What are the basic modeling elements for a mechanical system? Explain the function of each element.
-
You have $100,000 to invest in either Stock D, Stock F, or a risk-free asset. You must invest all of your money. Your goal is to create a portfolio that has an expected return of 10.5 percent. If D...
-
What kind of financial pressures can an LBO cause?
-
Explain why, in the reactions given in Fig. P18.68, p. 883, different stereoisomers of the starting material give different products. Fig. P18.68 oc., (only elimination product observed) H,C Br KOH...
-
When 1, 3, 5-trinitrobenzene [NMR: 9.1 (s)] is treated with Na+ CH3O-, an ionic compound is formed that has the following NMR spectrum: 3.3 (3H, s); 6.3 (1H, t, J = 1 Hz); 8.7 (2H, d, J = 1Hz)....
-
When 1, 3, 5-trinitrobenzene [NMR: 9.1 (s)] is treated with Na+ CH3O-, an ionic compound is formed that has the following NMR spectrum: 3.3 (3H, s); 6.3 (1H, t, J = 1 Hz); 8.7 (2H, d, J = 1Hz)....
-
Suppose the supply and demand schedules for cell phones are as follows: Price $2 Demand 11 Supply 1 $3 10 $4 9 $5 8 $6 7 $7 6 $8 5 $9 4 $10 3 $11 $12 2 1 10 23456700 8 9 11 The initial equilibrium...
-
Company B just paid an annual dividend of $.42 a share. The stock is selling for $18 a share and has a growth rate of 2.2 percent. What is the dividend yield, using the constant growth model?
-
Dooley, Inc., has outstanding $155 million (par value) bonds that pay an annual coupon rate of interest of 15.5 percent. Par value of each bond is $1,800. The bonds are scheduled to mature in 17...

Study smarter with the SolutionInn App


