Predict the major product for each reaction below: (a) (b) 1) KCN, HCN 2) LAH -? 3)
Question:
(a)
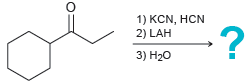
(b)
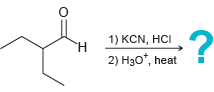
Transcribed Image Text:
1) KCN, HCN 2) LAH -? 3) H20 1) KCN, HCI 2) Hзо, heat
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a b 1 ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product for each reaction below: (a) (b) 1) [H'), HS 2) Raney Ni SH 1) [H*), HS' `H. 2) Raney Ni SH
-
Predict the major product for each proposed Diels-Alder reaction. Include stereochemistry where appropriate. (a) (b) (c) Ph 0 Ph
-
Predict the major product for each E1 reaction: a. b. eat Br H2SO4 eat
-
Include another public method in the Employee class from Pencil and Paper Exercise 3. The method should calculate an Employee objects new salary, which is based on the raise percentage provided by...
-
One of the most influential pieces of legislation was the Sarbanes-Oxley Act of 2002. This act greatly increased the transparency of financial disclosure in light of accounting scandals. See...
-
Use the solutions in Problem 29 to explain the outcome of each of the following collisions: (a) A moving truck colliding with a bike initially at rest. (b) A bike colliding with a stationary truck....
-
The stress-strain diagram of a material can be approximated by the two line segments. If a bar having a diameter of \(80 \mathrm{~mm}\) and a length of \(1.5 \mathrm{~m}\) is made from this material,...
-
Machinery purchased for $60,000 by Tom Brady Co. in 2010 was originally estimated to have a life of 8 years with a salvage value of $4,000 at the end of that time. Depreciation has been entered for 5...
-
How can Southeast Groceries Leadership encourage others to embrace and support change when rumors are flying? If you were head of HR at SEG, what would be your communication plan to employees for...
-
An aqueous solution containing 35 wt% MgSO 4 is fed to an evaporative crystallizer operating at 50F. The vapor generated is 20% by mass of the feed. The solution, which contains 23 wt% MgSO 4 , and...
-
Identify the reagents necessary to accomplish each of the transformations below: (a) (b) Me
-
Identify the reagents necessary to accomplish each of the transformations below: (a) (b) OH HO NH2
-
If f is continuous on [a, b] and what can you conclude about f ? SIS(x) | dx = 0.
-
Prepare journal entries to record the following transactions. Purchased $390 of supplies on credit. Completed $590 of work for a client on credit. Paid $390 cash towards the amount owed from...
-
Herself Health Medical Group sold a building for $170,000 in cash. The cost was $450,000 and the accumulated depreciation was $290,000. What is the actual cash cost?
-
Two taxpayers file as married filing jointly and have taxable income of $73,500, which includes a qualified dividend of $2,000? What is the amount of their tax liability for 2022?
-
Bill, a single taxpayer, earned 170,000 in salary, 20,000 in income from an S-Corp and 10,000 in dividends. What is the total FICA tax Bill will pay himself on all of his income? (Ignore any FICA tax...
-
Emerald Inc., a manufacturing company, has a sector that manufactures candles. It produced 21,800 units of candles in a month. It requires 0.00 hours per unit. The standard labor rate is $6 per hour....
-
Find the value of each variable for which the statement is true. [5 x+6] 4 y 2 4 - x w + 7| [0
-
A container holds 2.0 mol of gas. The total average kinetic energy of the gas molecules in the container is equal to the kinetic energy of an 8.0 10-3-kg bullet with a speed of 770 m/s. What is the...
-
Dimethylformamide (DMF), HCON(CH3)2, is an example of a polar aprotic solvent, aprotic meaning it has no hydrogen atoms attached to highly electronegative atoms. (a) Draw its dash structural formula,...
-
As noted in Table 3.1, the pKa of acetone, CH3COCH3, is 19.2. In Table 3.1 (a) Draw the bond-line formula of acetone and of any other contributing resonance form. (b) Predict and draw the structure...
-
Formamide (HCONH2) has a pKa of approximately 25. Predict, based on the map of electrostatic potential for formamide shown here, which hydrogen atom(s) has this pKa value. Support your conclusion...
-
A typical optic fiber has a thickness of only 6.00 x 10 nm. Consider a beam from a standard He-Ne laser that has a wavelength equal to 633 nm. Suppose this beam is incident upon two parallel slits...
-
What annual interest rate, r, is required to return $670 from $300 over 10 years? Give your answer as a percentage to two decimal places and assume growth compounds quarterly.
-
Collisions and elasticity In this problem, we'll analyze a one-dimensional collision between a car and a truck. You may find it helpful to use the elasticity parameter k, which is given by: = V1f-V2f...

Study smarter with the SolutionInn App


