Predict the major product(s) obtained when each of the following compounds undergoes hydrolysis in the presence of
Question:
(a)
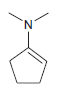
(b)
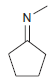
(c)
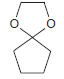
(d)
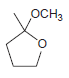
Transcribed Image Text:
z- z:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
a b ...View the full answer

Answered By

Mustafa olang
Please accept my enthusiastic application to solutionInn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group. For example, I created songs to teach my three-year-old campers the camp rules, but I gave my college student daily quizzes to help her prepare for exams.
I am passionate about helping students improve in all academic subjects. I still remember my excitement when my calculus student received her first “A” on a quiz! I am confident that my passion and experience are the qualities you are looking for at solutionInn. Thank you so much for your time and consideration.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many constitutional isomers are obtained when each of the following compounds undergoes monochlorination? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j)
-
Predict the product(s) obtained when each of the following compounds is treated with a mixture of nitric acid and sulfuric acid: (a) (b) (c) (d) (e) Br
-
Predict the product(s) obtained when each of the following compounds is treated with chloromethane and aluminum trichloride. Some of the compounds might be unreactive. For those that are reactive,...
-
Perform the indicated operations. (3k + q)
-
Develop 6 specific slides along with speaker notes that examine the importance of resource management and what role it plays on organizational success. Within your examination include the strategies...
-
Network models such as PERT and CPM are used a. to plan large and complex projects. b. to schedule large and complex projects. c. to monitor large and complex projects. d. to control large and...
-
The balance sheet of Oriental Rug Company at December 31, 2008, included the following stockholders' equity: During 2009, Oriental Rug completed the following selected transactions: Requirements 1....
-
Develop an outline (major headings and subheadings only) for a project management plan to create a Web site for your class, and then fill in the details for the introduction or overview section....
-
Evaluate [ sec r tan da +C
-
1.3 Determine the force P necessary for the equilibrium of a steel bar shown in Fig. 1.69. The diameters of the first, middle and the last segments of the bar are 30 mm, 25 mm and 30 mm respectively....
-
Glutaraldehyde is a germicidal agent that is sometimes used to sterilize medical equipment too sensitive to be heated in an autoclave. In mildly acidic conditions, glutaraldehyde exists in a cyclic...
-
Identify all of the products formed when the compound below is treated with aqueous acid: N- excess H,O+
-
In all the instances of molecules fitting together like "lock and key," you'll notice that the molecules involved are proteins. How are proteins able to achieve the specificity required?
-
Standing \(1.0 \mathrm{~m}\) away from a large mirror, you see an image of a flashlight in the mirror. If the image appears to be \(2.0 \mathrm{~m}\) behind the mirror, \(1.0 \mathrm{~m}\) vertically...
-
Consider the flat-plate heat transfer to be solved by the similarity approach. Show all the details leading to Eq. (18.15). What are the boundary conditions on \(\theta\) ? Extend your MATLAB code to...
-
Verify the integral obtained by Rayleigh and hence show that the velocity profile needs to have an inflexion point for instability. Show that a simple shear flow is stable. Hence viscosity is needed...
-
Analyze the Frank-Kamenetskii problem for the three standard geometries of slab, cylinder, and sphere. You will need to discretize the operators suitably for the cylinder and sphere. Plot the...
-
An infinitely long positively charged wire carries a uniform linear charge density \(\lambda\). A charged particle, carrying a charge \(q\), is placed at a distance \(2 d\) from the wire. (a) What is...
-
Describe the five components of an internal control framework.
-
Parkin Industries, a U.S. company, acquired a wholly-owned subsidiary, located in Italy, at the beginning of the current year, for 200,000. The subsidiary's functional currency is the euro. The...
-
Give the structure and stereochemistry of all products formed in each of the following reactions. (a) (b) trans-3-pentene Br H2O excess (solvent) ,o.roH cis-3-hexene BD3 solvent)
-
Give the structure of every stereoisomer of I,2,3- trimethylcyclohexane. Label the enantiomeric pairs and show the plane of symmetry in each a chiral stereoisomer.
-
Give the structure of every stereoisomer of I,2,3- trimethylcyclohexane. Label the enantiomeric pairs and show the plane of symmetry in each a chiral stereoisomer.
-
Sunland Company sells product 2 0 0 5 WSC for $ 7 0 per unit and uses the LIFO method. The cost of one unit of 2 0 0 5 WSC is $ 6 7 , and the replacement cost is $ 6 6 . The estimated cost to dispose...
-
Susan Wilson is opening a small flower shop that will focus primarily on delivery, though it will provide a small showroom for walk-in customers. A local florist is retiring and selling her small...
-
On January 1, 2019, Alsvin Company enters into a lease of equipment with Benni Company with a lease term of four years. The lease is properly accounted for as an operating lease. The rate Benni...

Study smarter with the SolutionInn App


