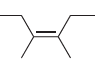
Predict the products that are expected when each of the following alkenes is treated with ozone followed
Question:
a.
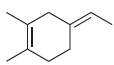
b.
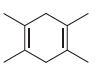
c.
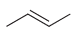
d.
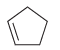
e.
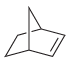
f.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a b ...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the products that are expected when each of the following compounds is heated with concentrated HBr. a. b. c. d.
-
Draw the products that are expected when each of the following amino acids is treated with ninhydrin: (a) l-Aspartic acid (b) l-Leucine (c) l-Phenylalanine (d) l-Proline
-
Predict the products formed when each of the following isotopically substituted derivatives of chlorobenzene is treated with sodium amide in liquid ammonia. Estimate as quantitatively as possible the...
-
Last year, Lena biked 330 miles. This year, she biked k miles. Using k, write an expression for the total number of miles she biked.
-
How should the analyst investigate the setup and tools to develop better methods?
-
1 percent of Volcanos shares were tendered. Philips then completed the merger without a shareholder vote. Before the merger concluded, three plaintiffs filed lawsuits, which were combined into a...
-
The Carnot cycle comprises four operational steps. These are (a) Two isothermals and two isochorics (b) Two isobarics and two isenthalpics (c) Two isothermals and two adiabatics (d) Two isothermals...
-
AutoIgnite produces electronic ignition systems for automobiles at a plant in Cleveland, Ohio. Each ignition system is assembled from two components produced at AutoIgnites plants in Buffalo, New...
-
The company has the following fixed costs: Product A, $613,000, Product B, $1,023,000, and common fixed costs of $372,800. Using the above information answer the following questions. What is the...
-
The following six transactions of Ajax Moving Company, a corporation, are summarized in equation form, with each of the six transactions identified by a letter. For each of the transactions (a)...
-
Draw molecules of ethanol and acetaldehyde.
-
Birds other than this gull species ignore the taped distress calls. a. What might account for the observed behavior in the other birds? b. Researchers switched young of one species of bird with those...
-
A supply of nitrogen is required as an inert gas for blanketing and purging vessels. After generation, the nitrogen is compressed and stored in a bank of cylinders, at a pressure of 5 barg. The inlet...
-
MargeSimpson Inc. is considering the two capital budeting projects with the following cash flows that have a WACC of 10%. How 'Bout A Pretzel Year What a Falafel -150,000 -150,000 1 30,000 90,000 2...
-
As employees health changes, they may miss work due to illness or injury increasing healthcare cost for employees and employers. Furthermore, employees may be less productive while at work due to...
-
Jones Company reports the following financial information for the current year: Net Sales $ 30,000 Cost of Goods Sold 9,000 Gross Profit 21,000 Operating Expenses 6,000 Operating Income 15,000...
-
Moerdyk & Co. is considering Projects S and L, whose cash flows are shown below. These projects are mutually exclusive, equally risky, and not repeatable. If the decision is made by choosing the...
-
Assume I have a stack of integer elements called st. What is going to be printed after I run the following statements? st.push(2); st.push(5); st.pop(); st.push(1); st.peek(); st.push(12);...
-
Evaluate the sum. i=1
-
Suppose that you could invest in the following projects but have only $30,000 to invest. How would you make your decision and which projects would you invest in? Project Cost $ 8,000 11,000 9,000...
-
There are two isomeric conjugate acids that produce the following base. Show the structure of each and explain how they produce the same base upon loss of a proton. :0: CH3-C=CH
-
When a is reacted with hydroxide ion, isomer b is formed. (a) Explain why b is favored over a at equilibrium. (b) Show the structure of the conjugate base of a and explain how this isomerization...
-
Compound c is a slightly stronger acid than compound d. The CH3O group has both an inductive effect and a resonance effect on the acidity of d. (a) Explain how the inductive effect of the CH3O group...
-
Athens Gas Station has figured out the weekly demand distribution for their gas sales. Each gallon of gas sold at the pump results in a profit of 10 cents/gallon and any lost sales results in a cost...
-
1. The maximum pressure an eardrum can withstand without rupturing is Pmax = 3.0102 Pa. Assume a sound is made at that pressure. If the density of air is, p=1.20 kg/m, and the speed of sound is 340...
-
52. A wave is sent down a string with a linear mass density of 1.600 x 10-4 kg/m. The wavefunction that describes this wave is: y(x,t) = (0.021 m)*sin{(2.00 rad/m)*x + (30.0 rad/s)*t} What is the...

Study smarter with the SolutionInn App


