Predict which side of the following equilibrium is favored, and explain your choice. Heat
Question:
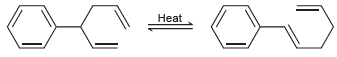
Transcribed Image Text:
Heat
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
The compound on the right has a bond in conjugation wi...View the full answer

Answered By

Mary Njunu
I posses Vast, diversified knowledge and excellent grammar as a result of working in ACADEMIC WRITING for more than 5 years. I deliver work in various disciplines with assurance of quality work. I purpose at meeting the clients’ expectations precisely. Let’s work together for the best and phenomenal grades.
4.90+
928+ Reviews
2551+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
At some temperature, a 100-L reaction vessel contains a mixture that is initially 1.00 mol CO and 2.00 mol H2. The vessel also contains a catalyst so that the following equilibrium is attained: At...
-
The following equilibrium is driven to the right if the reaction is carried out in the presence of maleic anhydride: What is the function of maleic anhydride? , ,
-
On which side of the equation do you expect each of the following equilibria to lie (left or right)? (a) (CH3)3 COH + K-OH- (CH3)3 CO-K+ + H2O. (b) CH3OH + NH3 CH3O- + NH-4; (pKa = 9.2). (c) (d) NH3...
-
Individual A is neither a trader nor a dealer for purposes of IRC 475. A purchases a single share of publicly traded Z stock for $1,000 on June 1, 2020. As of December 31, 2020, Z stock is trading at...
-
For the soil profile of Example 6.8 plot the total, neutral, and effective stresses with depth if the groundwater table is lowered 4 m below the ground surface.
-
Refer to problem 3-23. Find the variance and the standard deviation of hedge fund managers' income. x ($ millions) P(x) $1,700 ...... 0.2 1,500 ...... 0.2 1,200 ...... 0.3 1,000 ...... 0.1 800 .........
-
The maximum mechanical advantage is the reciprocal of PVs W slope. True or False
-
You have just been hired to the Senior Accountant position for Fitchtown LLC. During your first month in your new role, you will be completing a full accounting cycle for the month. You are the...
-
1. Two crates with treasure, a small one with mass m=110kg and a large one with mass M=329kg are connected by a massless rope and are on a frozen lake which you may assume to be frictionless. A rope...
-
On June 1, 2019, Kris Storey established an interior decorating business, Eco-Centric Designs. During the month, Kris completed the following transactions related to the business: June 1. Kris...
-
Predict the product for each of the following electrocyclic reactions: (a) (b) (c) (d) eat hv
-
When trans-3,4-dimethylcyclobutene is heated, conrotatory ring opening can produce two different products, yet only one is formed. Draw both products, identify which product is formed, and then...
-
Justify the statement that casting is most versatile forms of a mechanical process for producing components.
-
How would your answers change to exercises I through S if you were developing for a global marketplace?
-
How can you keep your customers engaged with your social media sites?
-
What do you think are three common mistakes that novice analysts make in navigation design?
-
What do you think are three common mistakes that novice analysts make in input design?
-
What do you think are three common mistakes that novice analysts make in output design?
-
Calculate y'. y = t 4 - 1/t 4 + 1
-
Ashlee, Hiroki, Kate, and Albee LLC each own a 25 percent interest in Tally Industries LLC, which generates annual gross receipts of over $10 million. Ashlee, Hiroki, and Kate manage the business,...
-
How flow might you convert geraniol into either ethyl geranylacetate orgeranylacetone? CO2E Ethyl geranylacetate CH2OH Geraniol Geranylacetone
-
How would you synthesize the following compounds from Cyclohexanone? More than one step may berequired. () CH2 CH2Br (a) (c) o CH2C6H5 CH2CH2CO2H (d) (e) (f) O
-
The two isomers cis- and trans-4-tert-butyl-2-methylcyclohexanone are inter-converted by base treatment. Which isomer do you think is more stable, and why?
-
Determine the slack / float of all activities.
-
Question Content Area Changes in current operating assets and liabilities indirect method Victor Corporation's comparative balance sheet for current assets and liabilities was as follows: Line Item...
-
Determine a value of m (if possible) for which f is continuous at x = 1. mx-3 if x <1 f(x)= 3x if x1

Study smarter with the SolutionInn App


