Propose a mechanism for each of the following transformations: a. b. H2SO4 Heat H2SO4 Heat
Question:
a.
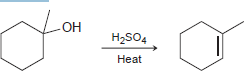
b.
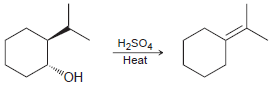
c.
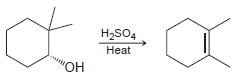
d.
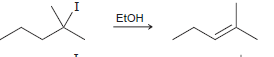
e.
Transcribed Image Text:
ОН H2SO4 Heat H2SO4 Heat "ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a b c ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose a mechanism for each of the following reactions: a. b. c. HO CH H3C CH CH2 CH3 H2SO4 H3C CH3 CH2CH3 CCl + HCCl peroxide
-
Propose a mechanism for each of the following reactions: a. b. CH3 CH-Br CH H20 CH H20 CH3 H3C CHCH CH3 CH3
-
Propose a mechanism for each of the following reactions: a. b. c. CH3 CH3 CH H2504 CH3 H2SO4 CH CH1 CHHBr_ CH3 CH
-
Using the Trust Services Principles and Criteria for the Online Privacy Principle, develop an online privacy policy for Alltel Stadium that could be posted on the stadiums website for customers to...
-
Question 1) How can the project manager determine the risk tolerance or risk appetite of the overall company? At what level of the company is this determined and how is it communicated? Question 2)...
-
What are the ranks of the credit cards in terms of their number and percentage of users from the most to the least popular?
-
Fill in the Blank. In the displacement method, the ___________ of elements is directly approximated.
-
Comprehensive problem including special order, outsourcing, and segment elimination decisions Huffman Corporation makes and sells state-of-the-art electronics products. One of its segments produces...
-
What the legal framework for cryptocurrencies in South Africa is. I need legal questions to help me get information to speak about the legal framework of cryptocurrencies in South Africa. I need all...
-
Jay Rexford, president of Photo Artistry Company, was just concluding a budget meeting with his senior staff. It was November of 20x4, and the group was discussing preparation of the firms master...
-
A new production technology for making vitamins is invented by a college professor who decides not to patent it. Thus, it is available for anybody to copy and put into use. The TC per bottle for...
-
In 2009 General Motors (GM) announced that it would reduce employment by 21,000 workers. What does this decision reveal about how GM viewed its marginal revenue product (MRP) and marginal resource...
-
In 1985, neither Florida nor Georgia had laws banning open alcohol containers in vehicle passenger compartments. By 1990, Florida had passed such a law, but Georgia had not. (i) Suppose you can...
-
SLMA Corp. for the last ten years, has earned and had cash flows of about P600,000 every year. As per the predictions of the company's earnings, the same cash flow would continue for the foreseeable...
-
England Corp. has identified the following costs at different activity levels for electricity for their manufacturing plant: Units Produced Electricity Cost 10,000 $1,700,000 15,000 $2,500,000 18,000...
-
Spring Toy Company sells musical pogo sticks to retail stores. At the beginning of the month Spring Toy had 590 pogo sticks in inventory that cost $2.50 each. They sold 1,440 pogo sticks for $33...
-
Altcom Company (the buyer) purchases $2,000 of merchandise on account from Bayside Tech Industries (the seller). The merchandise originally cost Bayside Tech Industries $1,500. Altcom Company found...
-
1.) How much of the office duties will you handle yourself? Contract out? 2.) Will you keep physical records or do everything electronically? 3.) How will you determine the appropriate fee structure?...
-
Evaluate the sum. -8) i=1
-
What are conversion costs? What are prime costs?
-
Which is stronger, the C = O bond in an ester (1735 cm1) or the C = O bond in a saturated ketone (1715 cm1)? Explain.
-
Carvone is an unsaturated ketone responsible for the odor of spearmint. If carvone has M = 150 in its mass spectrum and contains three double bonds and one ring, what is its molecular formula?
-
Carvone (Problem 12.39) has an intense infrared absorption at 1690 cm1. What kind of ketone does carvone contain?
-
If the nominal interest rate is 14%, and inflation is 4%, what is the real interest rate? 22.A bond investment yielded 8%. If inflation was 3%, what real return did the bond offer? 23.The following...
-
Bierce Corporation has two manufacturing departments--Machining and Finishing. The company used the following data at the beginning of the year to calculate predetermined overhead rates: Estimated...
-
Discuss how communication channels have changed over the years due to technology.

Study smarter with the SolutionInn App


