Propose a mechanism for the following transformation: OH H2SO4 Heat
Question:
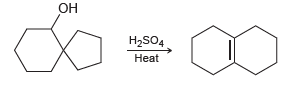
Transcribed Image Text:
OH H2SO4 Heat
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
H HOSOH OSO H ...View the full answer

Answered By

Niala Orodi
I am a competent and an experienced writer with impeccable research and analytical skills. I am capable of producing quality content promptly. My core specialty includes health and medical sciences, but I can competently handle a vast majority of disciplines.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw a mechanism for the following transformation: NaOH, heat
-
Draw a mechanism for the following transformation: 'CI Z Z
-
Draw a mechanism for the following transformation: . HCI -
-
What are the four different elements of advertising? Are all of them appropriate to an average retailer in most markets?
-
Here is the discussion on the topic of "Functional organizational conflict and a dysfunctional conflict and how a leader" Understandably leaders will encounter conflict, but the take away is that...
-
Analyze the following transactions and show their effects on the basic accounting equation, by preparing a table like the one in Exhibit 2-3: In Exhibit 2-3 a. Received $150,000 from investors buying...
-
Water flows over a flat surface at \(4 \mathrm{ft} / \mathrm{s}\), as shown in Fig. P6.42. A pump draws off water through a narrow slit at a volume rate of \(0.1 \mathrm{ft}^{3} / \mathrm{s}\) per...
-
Minnows Corporation began operations in 2014. At the beginning of the year, the company purchased plant assets of $1,800,000, with an estimated useful life of 10 years and no residual value. During...
-
IRAC answer to the question I was received about whether sam is criminally liable for assault for his actions against basil. Is this a good answer so far or is there anything I could change/doesnt...
-
At June 30 2017, the end of its most recent fiscal year, Green River Computer Consultants' post-closing trial balance was as follows: The company underwent a major expansion in July. New staff was...
-
(S)-1-Bromo-1,2-diphenylethane reacts with a strong base to produce cis-stilbene and trans-stilbene: a) This reaction is stereo-selective, and the major product is trans-stilbene. Explain why the...
-
Suppose that the last dollar that Victoria receives as income brings her a marginal utility of 10 utils while the last dollar that Fredrick receives as income brings him a marginal utility of 15...
-
The article "Analysis of Unwanted Fire Alarm: Case Study" (W. Chow, N. Fong, and C. Ho, Journal of Architectural Engineering, 1999:62-65) presents a count of the number of false alarms at several...
-
Name the steps (in sequence form) involved in the accounting cycle.
-
While balance sheet is like a snapshot, profit and loss account is like a moving picture. Explain.
-
What are nominal, real and personal accounts? Give four examples of each of them.
-
Extraordinary items warrant exclusion to judge true operating performance of a business enterprise. Elaborate. Give 3 examples each of abnormal losses and of abnormal gains.
-
Explain why decrease in current liabilities decrease cash and decrease in current assets increase cash.
-
Find the numbers at which f is discontinuous. At which of these numbers is f continuous from the right, from the left, or neither? Sketch the graph of f. 2 if x <1 f(x) = {3 x if 1 4
-
To help you become familiar with the accounting standards, this case is designed to take you to the FASBs Web site and have you access various publications. Access the FASBs Web site at...
-
We saw in Section 8.3 that addition of HBr to a terminal alkyne leads to the Markovnikov addition product, with the Br bonding to the more highly substituted carbon. How could you use 13C NMR to...
-
Identify the indicated sets of protons as unrelated, homotopic, enantiotopic, ordiastereotopic: (c) (a) (b) - C=C Hc CH (d) (e) (f) CH CH Br .
-
How many kinds of electronically nonequivalent protons are present in each of the following compounds, and thus how many NMR absorptions might you expect in each? (a) CH 3 CH 2 Br (b) CH 3 OCH 2 CH...
-
Can we justify the acquisition of Peloton Interactive Inc using NPV assuming the initial investment is 10% of the total market value of the company, 5% constant growth rate for the next three years...
-
Rivan Co. is considering a new three-year expansion project that requires an initial fixed asset investment of $2.18 million in a large building. The fixed asset applies three year MACRS for tax...
-
Mary would like to save $10,000 at the end of 5 years for a future down payment on a car. How much should she deposit at the end of each month in a savings account that pays 1.2%/a, compounded...

Study smarter with the SolutionInn App


