Propose a mechanism for the following transformation: 1) Excess LAH 2) H20 CH3
Question:
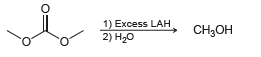
Transcribed Image Text:
1) Excess LAH 2) H20 CH3он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
JA HAH H ...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw a mechanism for the following transformation: NaOH, heat
-
Draw a mechanism for the following transformation: 'CI Z Z
-
Draw a mechanism for the following transformation: . HCI -
-
(2) Use Figure 10.2 shows a multi-degree-of-freedom (MDOF) model of three connected masses. Find the following for the MDOF system in Figure 10.2: Find expressions for the kinetic energy (T) and...
-
Quicksilver Delivery Service contracts to deliver Pete's Pizza Parlor's products to its customers for $5,000, payable in advance. Pete's pays the money, but Quicksilver fails to perform. Can Pete's...
-
Consider the following information for Mr. and Mrs. Di Palma: On June 10, 2022, they sold their principal residence for \($80,000\) and incur \($6,000\) of selling expenses. The basis of the...
-
Differentiate between the content and process theories of motivation. Which do you think are most useful to managers looking for ways to motivate their employees?
-
The records of Hollywood Company reflected the following balances in the stockholders equity accounts at December 31, 2010: Common stock , par $12 per share, 30,000 shares outstanding Preferred...
-
Five relevant types of power include: coercive, legitimate, reward, referent, and expert. What power do you think you would find most motivating in the workplace? Why? Include an example in your...
-
What characteristic does Ruby share with Smalltalk?
-
Show the reagents you would use to achieve the following transformation:
-
A carbocation is resonance stabilized when it is adjacent to an oxygen atom: Such a carbocation is even more stable than a tertiary carbocation. Using this information, propose a mechanism for the...
-
Time value of money calculations are used to determine the value of potential retirement benefits. If a person deposits $1,800 a year in a retirement account earning 6 percent for 20 years, what will...
-
Think about the different communities you belong and what, if anything you think needs to change in a positive way. 2. Decide on an action that you could take to make positive change in your...
-
Do you think using communication methods based on information technology (IT) is the best way for a large business to communicate with its suppliers? Justify
-
As an economy develops and becomes more integrated into the world economy how do its costs of production change and how well can manage them in both the short-run and long run.
-
What is the net income of sole proprietorships, partnerships and other unincorporated businesses?
-
A client with pneumonia has an order for Zosyn 250 mg diluted into 500 ml of NS. The medication is to run over 1 hour. The tubing gtt factor is 30 gtt per ml. How many drops per minute (gtt/min)...
-
Graph the hyperbola. Give the domain, range, center, vertices, foci, and equations of the asymptotes. |(y - 7)? (x- 4)2 = 1 36 64
-
Express these numbers in standard notation. a. 2.87 10-8 b. 1.78 1011 c. 1.381 10-23
-
Isoborneol (Problem 27.37) is converted into camphene on treatment with dilute sulfuric acid. Propose a mechanism for the reaction, which involves a carbocationrearrangement. C . H H2SO4 HO . H3...
-
Digit oxigenin is a heart stimulant obtained from the purple foxglove Digitalis purpurea and used in the treatment of heart disease. Draw the three-dimensional conformation of digitoxigenin, and...
-
What product would you obtain by reduction of digitoxigenin (Problem 27.39) with LiAlH4? By oxidation with pyridinium chlorochromate
-
Jackson, a self-employed taxpayer, uses his automobile 90% for business and during 2023 drove a total of 15,000 business miles. Information regarding his car expenses is listed below. Business...
-
Bel Air Flying Service started operations on December 1 , 2 0 . The trial balance in Figure 1 3 . 6 was prepared at month - end. Record the following adjustments in the General Journal, then make an...
-
Founded in 1960, the Arpeggio Music Company is a family-owned and operated firm specializing in the educational music market. Located in the U.S. Middle West, its products and services include...

Study smarter with the SolutionInn App


