Propose a plausible mechanism for the following transformation: D3o+ .D
Question:
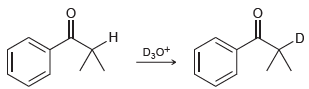
Transcribed Image Text:
н D3o+ .D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
H...View the full answer

Answered By

SANDESHDEV PRASANNA
My name is sandesh, I'm a Math tutor from past 9years teaching math in a simple way which make students love subject with more understanding and easier way to crack the competative exams.
Thank you
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw a plausible mechanism for the following transformation: , [H2SO4]
-
Draw a plausible mechanism for the following transformation: -NH2 [H,SO] NH2 [-H,0]
-
Draw a plausible mechanism for the following transformation ,*
-
With a fixed cost of $100 per order, Nathan decided it was vital to get his money's worth. His monthly demand for energy drinks was 10,000 bottles and holding cost was estimated at 20% of unit cost....
-
The daily grind uses a specialty brand of Kona coffee beans costing $8 per pound. Each pound of coffee beans produces 256 ounces of coffee. Coffee is sold in three sizes: a small cup holding 8...
-
Provide two examples of a tax preparer penalty.
-
Explain the meaning of push and pull promotional techniques. How can each help the salesperson to plan sales more effectively?
-
Orchard Company has monthly salaries of $10,000. Assume Orchard pays all the standard payroll taxes and no employees have reached the payroll tax limits. Journalize the accrual and payment of...
-
Blue Ridge Hiking Company has an estimated sales price of theheadlamp is RM19.99 per unit. The variable cost per unit isRM10.29. Blue Ridge Hiking Company has annual fixed costs totallingRM109,500....
-
Which of the following is NOT a uniquely new trait of cloud computing problems compared to the previous generations of distributed systems problems? Servers coordinating with each other Large amounts...
-
Use the following information to analyze the financial and inputs/outputs of the organization from 2015 to 2016: Financial Information 2015 2016 Results Results $1,372,000 $1,416,400 Netsales Cost of...
-
Calculate H o R at 675 K for the reaction 4NH 3 (g) + 6NO(g) 5N 2 (g) + 6H 2 O(g) using the temperature dependence of the heat capacities from the data tables. Compare your result with H o R at...
-
Write the net ionic equation for the reaction of the strong electrolytes nitric acid and potassium hydroxide.
-
Since the year 2000, there has been a significant trend in the application of the death penalty in the United States. Describe the trend and provide some of the reasons for the change.
-
how Employee Training Program worked in other businesses by describing your own experience in a credible way or by providing examples of business leaders who have faced similar challenges in similar...
-
Consider the possible ways that students demonstrate collaboration skills through teamwork in the classroom. What are some strategies that teachers could use to encourage this behavior and provide...
-
1. A construction contract called for oak paneling in certain rooms. Because oak was unavailable, a subcontractor installed birch panel. The prime did not notice this deviation. Oak and birch...
-
The statues or acts of province ( and Canada) set out the law on a particular topic. What indicates the guidelines to carry out the law set out in the act or statue, how are they referred to ?
-
Use the determinant theorems to evaluate each determinant. 2 -1 3 4 10 4 5 6.
-
At 31 December 20X9, the end of the annual reporting period, the accounts of Huron Company showed the following: a. Sales revenue for 20X9, $ 2,950,000, of which one- quarter was on credit. b....
-
Tyramine is an alkaloid found, among other places, in mistletoe and ripe cheese. How would you synthesize tyramine from benzene? Fromtoluene CH2CH2NH2 Tyramine
-
How would you prepare the following compounds from toluene? A diazonio replacement reaction is needed in someinstances. (a) (b) (c) NH2 CH2NH2 H
-
Reaction of anthranilic acid (o-amino benzoic acid) with HNO2 and H2SO4 yields a diazonium salt that can be treated with base to yield a neutral diazonium carboxylate. (a) What is the structure of...
-
The Presentation Layer ER Diagram shown in conjunction with part a below is focused around students who participate in sports and also enroll in classes. Please base your answer to this question on...
-
Establishment Industries borrows $1,040 million at an interest rate of 7.2%. It expects to maintain this debt level into the far future. Establishment will pay tax at an effective rate of 38%. What...
-
The current price of one share of XYZ stock is 56.50. A long straddle (one long call and one long put) on the stock with a strike price of K and one year to expiration has a total cost of 12.00. The...

Study smarter with the SolutionInn App


