Provide an IUPAC name for each of the following alcohols: a. Br Br
Question:
a.
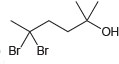
b.
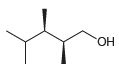
c.
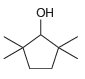
d.
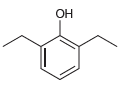
e.
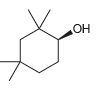
Transcribed Image Text:
"ОН Br Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
a 5 5dibromo2methylhexan2ol b ...View the full answer

Answered By

John Aketch
I am a dedicated person with high degree of professionalism, particularly in academic writing. My desire is to is to make students excel in their academic endeavor.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Provide an IUPAC name for each of the following alcohols: a. b. c. d. e. HO. Br Br
-
Provide an IUPAC name for each of the following compounds. a. b. c. d. e. I
-
Give the IUPAC name for each of the following hydrocarbons. a. b. c. d. , , CH3 CH3 CH3CCH2CH2CH2CCH3 CH3 CH3 CH CH2CHCH2CH2CH2CH3 CH2CH2CH3 CH3 CH CHCHCH2CH2CH2 CH3 CH2CH3
-
Aria Perfume, Inc.. sold 3,210 boxes of white musk soap during January of 2016 at the price of $90 per box. The company offers a full refund for any product returned within 30 days from the date of...
-
Prove Markov's Inequality: If X is any random variable and a > 0, then Pr( |X| a) E( |X| )/a. Show how this inequality can be applied to Theorems 5.2 and 5.3.
-
How can you evaluate the effectiveness of training?
-
Richard Hernstein, who has audited the financial statements of the Century Group, Inc., a publicly held company, for the year ended December 31, 1999, has been asked to perform a limited review of...
-
Marks Consulting experienced the following transactions for 2016, its first year of operations, and 2017. Assume that all transactions involve the receipt or payment of cash. Transactions for 2016 1....
-
The following data relate to the operations of Shilow Company, a wholesale distributor of consumer goods: Current assets as of March 31: Cash Accounts receivable Inventory Building and equipment, net...
-
Given a database of the results of an election, find the number of seats won by each party. There are some rules to going about this: There are many constituencies in a state and many candidates who...
-
The institution of teacher tenure is meant to a. Ensure job security for teachers with 10 years of experience. b. Ensure that teachers do not get fired for political reasons. c. Allow teachers to...
-
Determine the electron configuration for each of the following ions: a. A carbon atom with a negative charge b. A carbon atom with a positive charge c. A nitrogen atom with a positive charge d. An...
-
At a Als Appliances, 1/4 of those purchasing appliances purchase a dishwasher. Determine the probability that (a) None of the next four people who purchase an appliance at Als Appliances purchases a...
-
Consider two descriptive facts. First, the poorest countries in the world are disproportionately located close to the equator. Second, the civilizations that were richest in 1500 are among the...
-
Why did big business get bigger in the US than in other industrialized countries (e.g., Britain and Germany) during the 2d Industrial Revolution? How did the US differ from other countries such as...
-
$400,000 for a new technology.The lender charges them 36% annually with monthly compounding.The agreement calls for no payment until the end of the first month of the 5th year with equal monthly...
-
16. Brenda's Balloon Shop is a price taker, and the table below shows its costs of production. Use it to answer the following questions: Output (balloons Total Cost (dollar per hour) per hour) 0 4 1...
-
To solve this model economy, we reformulate the competitive equilibrium into the social planner's problem. First of all, in social planner's problem, all markets must clear, and thus N = Nd = N, and...
-
Solve each system of equations. State whether it is an inconsistent system or has infinitely many solutions. If the system has infinitely many solutions, write the solution set with y arbitrary. 2x -...
-
Find the numerical value of each expression. (a) sech 0 (b) cosh -1 1
-
Alcohols can act either as weak acid or as weak bases, just as water can. Show the reaction of methanol, CH 3 OH, with a strong acid as HC1 and with a strong base such as NA + - NH 2 .
-
The O ? H hydrogen in acetic acid is much more acidic than any of the C ? H hydrogens. Explain this result using resonance structures. Acetic acid
-
Write the products of the following acid-base reactions: (a) CH3OH + H2SO4 ? (b) CH3OH + NaNH2 ? (c) CH3NH3 + C1- +NAOH ?
-
Find the complete solution of the linear system, or show that it is inconsistent. (If the system has infinitely many solutions, express your answer in terms of t, where x = x(t), y = y(t), and z=t....
-
1. What is the difference between invitation to treat and offer? 2. Explain the statement "acceptance must be absolute and unqualified." Question 3 Ali goes to the convenience store to buy a can of...
-
The firm sold one of its fixed assets just as the year ended. Would this imply that operating cash flow for the year was a larger figure, a smaller figure, or would have no effect on its operating...

Study smarter with the SolutionInn App


