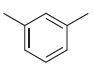
Provide at least five different acceptable IUPAC names for the following compound.
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
a Metaxylene b 1 3...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give IUPAC names for the following compounds: CH CH3CH2CH=CCH2CH3 (b) (a) CH CHCH-H CHCH2CH2CHCHCHCH2CH3 CH CH (d) (c) CHCHCHCHC CH
-
Give IUPAC names for the following cycloalkanes: (c) CH2CH2CH3 (b) CH (a) CH CH3 (f) Br (e) C CH-CH (d) CHICH3)2 "CH C(CH3)3 Br
-
Give IUPAC names for the followingcompounds: (b) (a) The three isomers of C5H12 CH CH;CH-CHH CH (d) (c) CH CH (CH)2CHCH2CH3 (CH3)3CH-H2C CH
-
A rectangular field will have one side made of a brick wall and the other three sides made of wooden fence. Brick wall costs 20 dollars per meter and wooden fence costs 30 dollars for 3 meters. The...
-
The dry density of compacted sand is 1.87 Mg/m3 and the density of the solids is 2.67 Mg/m3. What is the water content of the material when saturated?
-
The amount of mineral water filled in 2-liter bottles by an automatic bottling machine is monitored using a control chart. The actual contents of random samples of 4 bottles collected on 8 different...
-
What are the effects of air infiltration in condensers ?
-
The town of Millbridge has just agreed to pay a pension to the town clerk. The pension will be $ 40,000 per year for the next 20 years. Dwight Ives, the town manager, has decided that the town should...
-
1. Consumers often make purchase decisions based on what they have purchased before. What can marketers do to help influence a shift in behavior for frequently purchased, low-elaboration goods like...
-
The following table lists the distribution of degrees in psychology conferred by degree-granting institutions, by sex and year. These and similar data are reported by the National Center for...
-
In Chapter 9, we saw that meta-chloroperoxybenzoic acid (MCPBA) is a peroxy acid commonly used to convert alkenes into epoxides. Recall that peroxy acids have the following structure a) Draw the...
-
Which water pollution law regulates industrial waste discharge into a river?
-
(a) The dipole moment of trans-1,3-dibromocyclobutane is 1.1 D. Explain why a nonzero dipole moment supports a puckered structure rather than a planar structure for this compound. (b) Draw a...
-
Suppose the interest on Russian government bonds is 7.9%, and the current exchange rate is 28.6 rubles per dollar. If the forward exchange rate is 29.1 rubles per dollar, and the current U.S....
-
Arden Corporation is considering an investment in a new project with an unlevered cost of capital of 8.5%. Ardens marginal corporate tax rate is 37%, and its debt cost of capital is 4.8%. a. Suppose...
-
Constant growth Blix Net services provides dedicated server facilities for small businesses. The company paid per share dividends of 1.21, 1.37, 1.46, 1.61, 1.76, and 1.87, respectively, in the six...
-
Rebecca is interested in purchasing a European call on a hot new stock, Up, Inc. The call has a strike price of $99 and expires in 92 days. The current price of Up stock is $119.16, and the stock has...
-
Peter Brierley, based in Singapore, wishes to determine how long it will take an initial investment of SGD 10,000 to double. a. If Peter earns 7% annual interest on his investment, how long will it...
-
Find the most general antiderivative of the function. f(x) = (x 5)?
-
Use the graphs of f and g to graph h(x) = (f + g) (x). To print an enlarged copy of the graph, go to MathGraphs.com. 1. 2. y 24 8. 2. -2 -2 4 6
-
Orient each of the following drawings so that the lowest-priority group is toward the rear, and then assign R or Sconfiguration: (b) (a) (c) 3
-
Assign CahnIn gold-Prelog priorities to the following sets of sub-stituents: (a) H2, H)2, ICH3)3, CH2CH3 -H3)2. -C(CH3)3, -CH2CH3 () , CH2, (CH3)3 -CH=CH2, -(CH)3, () ,H , 3, H20CH3, H-H...
-
Assign R or S configurations to the chirality centers in the followingmolecules: CI H la) (c) (b) H
-
A one cubic foot sample of a borrow-source clay weighed 88 lbs. If the specific gravity of solids was measured as 2.70, and the clay was found to be 10 percent saturated, determine the water content...
-
Evaluate the use of novel catalytic systems, such as structured catalysts, catalytic distillation, and multifunctional catalysts, in achieving process intensification, discussing the effects on...
-
Find the minimum tractive effort required for vehicle to maintain 70mph speed at 5%upgrade through an air density of 0.002045 slug/ft^3. Show all steps and unit conversion please Problem 2:...
Estuaries The Handbook Of Environmental Chemistry 5 H 2006 Edition - ISBN: 9783540002703 - Free Book

Study smarter with the SolutionInn App


